Weight loss engages a “starvation response” that acts to regain the lost weight, and this is a key reason why weight loss is difficult and often temporary. This starvation response includes an increase in the drive to eat and a decrease in calorie expenditure. Using a clever study design, Kevin Hall and colleagues recently quantified the contribution of eating drive to this starvation response. The results suggest that increased eating drive is the primary way in which the starvation response opposes weight loss.
Countless controlled trials have shown that weight loss is difficult, regardless of the approach. Not only is it unlikely that a person with obesity will lose all of his excess fat, but in most cases even moderate losses are regained over time. A key reason for this is that weight loss decreases the circulating concentration of the hormone leptin, activating a negative feedback process that I call the “starvation response”, which I explain in greater detail in my book. This response attempts to bring more calories into the body, and curtail the flow of calories out of the body, thereby refilling fat stores. The starvation response includes an increase in the drive to eat (hunger, food seductiveness) and a decrease in calorie expenditure (slowed metabolic rate and physical activity calorie expenditure).
The “calories out” side of this equation has been fairly well characterized, yet we don’t know nearly as much about the “calories in” side due to the challenges inherent in measuring it. A new paper from Kevin Hall’s research group asks the important question, how strongly does the increased drive to eat counter weight loss? To do this, they used a clever study design in which weight loss was covertly induced.
The study
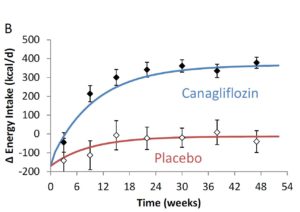
Hall’s team used data from a one year randomized, placebo-controlled trial of a drug called canagliflozin in 242 people with type 2 diabetes and obesity. Canagliflozin causes substantial loss of glucose via the urine and is used to improve blood glucose control in diabetes. This loss of glucose is roughly 90g per day, amounting to about 360 Calories.
Importantly, since this was a double-blind trial, neither the investigators nor the subjects had direct knowledge of who was receiving the drug. The study involved a long-term, covert perturbation of energy balance that “largely bypasses the volition of the subjects”, which makes it an unusually pure test of the brain’s response to changes in body weight.
Using previously validated mathematical models, and measured body weight changes, Hall’s team estimated the calorie intake of the canagliflozin and placebo groups over the course of the one-year trial. They then used the resulting output to estimate how calorie intake, calorie expenditure, and the drive to eat change with typical weight loss, using two years of data from a commercial weight loss program (Weight Watchers).
The results
The canagliflozin group gradually lost weight over the course of the trial, eventually plateauing about 8 pounds (3.5 kg) lighter than at the beginning. In contrast, the placebo group lost about two pounds (just under 1 kg).
Hall’s model estimates that as the canagliflozin group lost weight and the starvation response kicked in, their calorie intake gradually increased by about 350 Calories per day, eventually halting their weight loss. The model estimates that for each 2.2 pounds (1 kg) of body weight lost, the brain ramps up the drive to eat by about 100 Calories per day in an attempt to regain the weight!
With this information in hand, the researchers were able to gain insight into what happens during typical weight loss due to diet and lifestyle change. Data from a two-year commercial weight loss trial showed that participants experienced their maximum weight loss at 6 months, then gradually regained over the following year and a half.
Plugging these numbers into Hall’s model, they estimate that the initial calorie deficit was about 700 Calories per day, but that this rapidly rebounded, and had almost returned to baseline by 9 months. This might suggest that the participants just stopped trying, but in fact this is unlikely to be the case, because they were still weight reduced at that point and therefore their brains were still experiencing a heightened drive to eat (see the line below labeled “homeostatic drive to eat”).
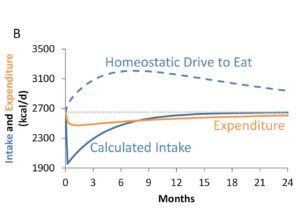
The researchers were actually able to quantify the effort the participants were putting into their diets by calculating the difference between actual calorie intake and the estimated drive to eat. The results suggest that even though people were already almost back to their baseline calorie intake by 9 months, and already beginning to regain weight, they were nevertheless expending considerable effort to stop themselves from eating substantially more than their baseline intake (to the tune of about 600 Calories per day).
Gradually, as their body weights rebounded over the last year and a half of the trial, their effort level declined toward baseline.
Discussion
This study has a notable strength, which is that energy balance and body weight were perturbed without the participants’ knowledge. We can be confident that the effects the researchers observed had nothing to do with the participants’ conscious feelings or behaviors around dieting and weight loss. They were the result of nonconscious biological processes.
The study also had at least two notable weaknesses. The first is that the participants in the first trial had type 2 diabetes, which means that we can’t know how well the results would apply to people without diabetes. I’m not aware of any reason why they wouldn’t apply, and the results are consistent with what we know about weight regulation in people without diabetes, but we still have to be cautious. The second weakness is that the trial perturbed calorie balance via a specific mechanism– the excretion of blood glucose in the urine. It’s theoretically possible that the results would have been different if the drug had caused the participants to excrete fat. I don’t think this is very likely, but this question might be tractable by studying randomized trials of Olestra, the fat substitute that gets excreted in feces. Or perhaps by studying liposuction patients.
There are several interesting implications of this study. The first is that the data support our current understanding of how body weight is regulated. Even when energy balance was perturbed without the participants’ knowledge, a powerful starvation response occurred that favored the regain of lost weight. The data support the concept that a sort of body weight “set point” is defended against changes, particularly changes in the downward direction.
A second implication is that between the two arms of the starvation response– the increased drive to eat and the decreased calorie expenditure– the former is by far the most influential. In other words, the primary way in which the brain opposes weight loss is by increasing the biological drive to eat. The data also give us a quantitative estimate of this drive: for each 2.2 lbs (1 kg) of weight lost, the drive to eat increases by about 100 Calories per day*. This is even stronger than I would have predicted.
A third implication– which I think is the most novel of the study– relates to the dynamics of the heightened drive to eat that people experience when they diet, and how this undermines weight loss efforts. After a person loses weight, the biological drive to eat can be so high that they have to exert considerable effort just to prevent themselves from overeating substantially. Even though it seems like they’re no longer adhering to their reduced calorie regimen, they may still be trying hard to eat fewer calories– and succeeding, relative to the amount their brain “wants” them to eat.
The amount of effort that people put into a diet does slowly decline over time however, and as this effort recedes, the biological drive to eat takes over and weight comes back. It’s hard to fight the starvation response forever.
This study strengthens my conviction that the brain’s starvation response has to be managed for long-term weight loss success– and particularly its impact on the drive to eat. Rather than pretending like the starvation response doesn’t exist, as most weight loss approaches do, a better method may be to try to dampen appetite and/or passive calorie intake sufficiently to counterbalance the heightened drive to eat. In other words, find ways to keep the brain happy at a lower calorie intake rather than using willpower to continually struggle against the biological drive to eat.
* I don’t think these results can tell us whether or not the relationship is linear, e.g. if 1 kg of weight loss causes an increase in the drive to eat by 100 Calories, would a 5 kg loss cause an increase of 500 Calories, 10 kg, 1000 Calories, etc.? It’s possible that there is a plateau at some point where the response reaches its maximum strength. Another way of saying this is that this simple rule of thumb may not necessarily apply over the entire range of weight changes. We would need more research to explore this possibility.


Very good point about the feedback factor kp being linear over wider ranges. I’m sure Hall’s feedback model would still work with kp a function of (BWi – BW0), i.e., the ΔEI(t) feedback would be proportional to (BWi – BW0)^2.
By the way, a “P+I” (proportional plus integral) feedback function doesn’t really make any sense, since it will always drive the (BWi – BW0) term to zero (always perfect regain of weight). That part of the paper could be taken out.
There is a theory that the response only occurs under a specific caloric level. From what I’ve read for a 150 pound male that level is 1600 calories. If dietary intake is kept above that amount but still under expenditure the starvation response doesn’t kick in. The weight loss is very slow but study. Unfortunately, most people aren’t satisfied dropping a pound or two a month and push their bodies to levels that force it to push back.
So do you suggest that we should use refeeds to restore leptin levels and thus remove the ‘starvation response’? Would this then allow us to change our bodyfat ‘set point’?
If so, how would you approach this?
As I lost weight my desire to eat increased, and I could not control hunger without eating more. At 215 lbs (obese Type II diabetic) I could eat 1500 kcal/day and not feel hungry. At 190 lbs 2000 kcal/day left me ravenously hungry by 5 AM every morning. In order to maintain a weight loss rate of 2 lbs a week and eat those extra 500 kcal/day I had to start expending a lot more energy. It was not possible to remain sedentary and continue to lose weight. My weight loss continued to 165 lbs, where I gave up.
10 years later I have maintained 40 of the 50 lbs lost. I have intentionally regained 10 lbs to replace muscle wasting (shoulders/arms/thighs) caused by the initial loss, and have not regained waistline circumference. To maintain the loss I cannot become sedentary or I experience immediate weight regain. And I am still ravenously hungry at 5 AM every morning even though I now eat 2200-2300 kcal/day. I can ease the morning hunger pangs by getting up and moving around, and by eating a small breakfast. Peanut buttered toast and coffee has worked the best for the last 10 years.
Eating an added 100 kcal per kg weight loss would probably end my morning hunger. At my current BMR of 1600 this would put me at 3600 kcal a day to eat. I would have no problem eating that much, but I would also have to spend 12 hours a day walking to maintain my weight.
While eating restriction is effective for weight loss, I don’t think it’s reasonable to expect permanent eating restriction to 1500 kcal/day to maintain weight. Our digestive systems run well at a reasonable midpoint and higher (Hazdas gorging on honey for instance) and not so well on daily minimums. I’ve found that it is easier to trick the brain with increased calories-out than reduced calories-in for weight maintenance.
http://www.nytimes.com/2012/01/01/magazine/tara-parker-pope-fat-trap.html
Hi thhq, you said you have been diabetic, how is your blood sugar reading , now that you are on a carbohydrate rich diet? Which
I assumed ,you eat carbs and deplete all your glycogen by walking by not being sedentary.
It stays around 100 fasting. The diabetes was diagnosed on high A1C (around 8 if I remember, now in the 5’s) and fasting blood glucose of 200 on repeated tests. My Dr. asked me what I had done to screw up my diet. I told him frosted miniwheats and raisin bran. He recommended peanut buttered toast instead. It’s been a go-to breakfast ever since. It kills my appetite.
Having a blood glucose monitor and lots of strips allows many experiments. In the first few months, I found that I could spike 200 post prandial with a piece of pie or with a relatively small serving of sushi rice.
But you’re obviously having to exert great mental expense to keep the weight off. I’m down about 40 pounds of so still after 2 years from my highest weight but it’s mentally expensive. More proof to the fact of what Stephan is saying is sadly right. Nobody is arguing that you can’t force weight down; there’s nothing against the law of physics that says you can’t lose fat, but that’s not to say the brain won’t respond with starvation like symptoms from reduced leptin causing increased desire, urges etc. which require great mental effort to overcome.
After 10 years my weight maintenance is a trained response. The most difficult part is that calories-out takes time, typically 2-3 hours a day. This is easier since retirement. When I was working, I walked 3-4 miles each way to and from work, at lunch and on breaks, to maintain 10 miles a day.
Hi, Stephan.
Interesting work by Hall’s group.
How do you think this reasoning applies to daily intermittent fasting?
Because in intermittent fasting we might have a calorie deficit that is even larger than in conventional weight-loss diets. But, based on the scientific literature, intermittent fasting doesn’t seem to have a big impact on overall hunger. Nor on food intake or on participant’s ability to keep loosing weight for the duration of the studies (which are usually not very long in duration, but still).
Thanks.
Has there been any study on the effect of long term weight loss by using stimulants. which I believe suppresses hunger by some means.
I’ve lost 25 lbs over the last 2 years eating a high starch, low fat McDougall Plan diet. My experience is that I will lose a few pounds, then rest there for a few months. When I decrease my fat intake and increase starches, I will lose a few more pounds. All weight loss has been slow and unsteady and I have had no weight gain. And the more starch I eat, the more satiated I am and the more weight I lose. I haven’t counted calories. I eat oats, berries, fruit, brown rice and other whole grains, potatoes, sweet potatoes, legumes, a few nuts and seeds, and a variety of veggies. I eat huge piles of food! My friends and family are amazed at how much I can eat. I am 5’6″ and now weight 140 lbs.
Some days I am very hungry so I eat more potatoes and sweet potatoes for a few days until my hunger stabilizes. I have to eat lots of food every day otherwise I start to “desperately seek calories” from any source (chips, chocolate, etc.). I also talk to myself about what is going on. When I’m losing weight I reassure my brain that everything is ok, we are not in a famine, and the kitchen is full of food. I lose a few pounds according to the bathroom scale, then everything settles down.
Most of the time over the past 2 years I have not thought about food much. But when I start thinking about food obsessively I know I’m losing fat and that’s when I begin my reassuring self-talk and up my intake of potatoes.
My goal is good health. The weight loss has been a nice extra benefit. I wasn’t really trying to lose weight. I believe that I am now in a position to maintain this weight forever. I completely changed what I eat and I have no other food in my house. And I have the tools to help myself over the rough spots.
So far, so good!
What do your macros look like? That is % of daily calories from fat, protein, carbohydrate?
As a thin person I have always curbed my hunger by drinking coffee which suppresses my appetite. Is it possible to have people trick their starvation response somehow, like with an appetite suppressant, or is that unhealthy?
I am sure long term stimulate use is unhealthy but an EC stack is probably the most effective weight loss tool I have ever used. Ephedrine + Caffeine – crushes appetite but also causes weight loss by some other mechanism. Back in my younger days, even on the same macros and calories, adding ephedrine caused a huge increase in weight loss.
I, too, am interested in the use of “refeeds” in turning off this starvation response. Any data or theories regarding that?
Hi Kevin,
I know some people have suggested that refeeds can help with the starvation response. I’m not aware of scientific evidence that speaks to that hypothesis. I’m skeptical but I don’t really have a basis for evaluating the idea.
Hi Stephan
I’ve restricted my net time for a while & I come back to find your book’s out … Congratulations.
I’ll be reading it soon.
Bob Johnson:
> Has there been any study on the effect of long term weight loss by using stimulants
If you count DNP as a stimulant, I suppose that’s 100% effective. The body does not seem to do anything to counter the adaptations (aka toxic effects) that DNP causes.
It’s usually NOT classed as “stimulant”, which usually means “a central nervous system stimulant” like ephedrine or amphetamines.
Oh … Stephan, I’m curious if you’ll be revising your thinking on conclusions from Dr. Wansink’s work in light of the recent … “happenings”.
Hi Sanjeev,
I have cited his work occasionally in my writing, including in my book. Recent events have definitely caused me to question his work. I won’t be relying on it until further notice. But I’m open to the possibility that some of it is still useful, if that can be sorted out.
But the main thing I use his work to support is the idea that convenience influences food choice and intake, which is not an especially controversial concept. Omitting his research won’t have any impact on my my views about eating behavior.
I didn’t answer your actual question, which was about studies, not about effectiveness
> Has there been any study on the effect of long term weight loss by using stimulants
For DNP There were studies way back, so questions about control remain. They were as well controlled as studies of the time I suppose.
But nothing recent, and very little long term because of the deaths.
> cirelo: As a thin person I have always curbed my hunger by drinking coffee which suppresses my appetite
(you may be an exception, BUT) … I really doubt it.
http://www.ncbi.nlm.nih.gov/pubmed/6871070
Steve Novella likes to say that after 4 weeks of heavy coffee use, 2 to 3 times per day, coffee’s biggest effect on the body is to prevent withdrawal symptoms.
There’s similar data (to the heart rate & blood pressure data) for how quickly people wake up, the focus & attention effects, diuresis and other effects of caffeine.
There are, I believe, effects on liver & muscle tissue that continue for a long time but still do get weaker with time.
That one study used high doses.
When Dr. NOvella talked about the effects he talked about many other studies using lower doses.
How can you “win” through the increased satiety (or any other) diet, when your bodyweight will either return to baseline – and you may still need more effort than if you just did no intervention to stay on the initial weight – or you’ll continue to fight starvation response until the weight does return to baseline? Why not just throw in the towel and focus on sleep, stress and an adequate amount of movement? It seems like more effort on diet means worse situation in 2 years.
I’d be interested to know the role of satiety in the dynamic explored in the study. For example, if lower calorie, high fiber foods (e.g., raw vegetables, cooked root vegetables) could “trick” the starvation response. Or can the brain not be fooled in the end and calories prevail regardless of how full one might feel.
That seems likely given the anecdotal reports of people not being able to eat enough plain potatoes to maintain weight. Potatoes are the highest satiety food measured in the Holt study and also (if not relatedly) are very low food reward.
That said, designing a diet that will result in calorie reduction without calorie counting is one thing. The hard part is still long term compliance.
to John’s point, I think satiety and food reward are two sides of the same coin. Oreo cookies would likely score low on ‘satiety’, partly because the fact of their being delicious (having extreme reward value) results in a greater drive to consume before reaching satisfaction.
Regarding ‘tricking’ the starvation response, I think Stephan’s main view is that the extent to which you’re tempted to take another bite is dominated by the food reward value of the food. So it follows that, after losing some weight, you need to surround yourself with foods that are at a low enough reward value that you spontaneously achieve a desirable body weight. Some people can have a house full of cookies and chips and maintain a healthy body weight. I cannot! But if I have a house full of whole foods, I spontaneously keep a healthy body weight (down 35 lbs), due to their reward value. Is this what you mean by ‘tricking’ the starvation response?
The initial caloric deficit of 700 cal/day seems insanely high to me, and I’m not surprised the brain would freak out and seek food. I’d be curious if these effects still apply at a more moderate deficit of 100-200 calories. Anecdotally I’ve been able to keep weight off for 10 years using this approach.
Hi Darya,
700 kcal/d is a substantial deficit but not that far from what people impose when they go on typical weight loss diets (~500 kcals). Calorie intake for the “average” person with obesity is in the 3,000 kcal/d range, so 700 kcals is a reduction of about a quarter.
To the best of our current knowledge, the energy homeostasis system doesn’t care that much whether fat loss is fast or slow, it mostly cares where body fatness currently is relative to the body fat “setpoint”. So whether a person loses 20 lbs quickly or slowly shouldn’t make much difference. That said, the location of the setpoint depends in part on diet quality and lifestyle, so it’s possible to lose fat comfortably/sustainably and without triggering a “starvation response”, at least for many people.
I cover all this in greater detail in my book, which I’d be happy to send you (I tried contacting you through your website a while back to send you a copy). Just send me a message at s.guyenet at gmail dot com.
Stephan: is there any data on how to permanently eliminate the starvation response after weight loss? I understand that you can choose low reward value foods to compensate for the SR, but is there any way to eliminate or reduce it? Thanks.
Hi Jeff,
I discuss this at length in my book, but here are a few quick thoughts. First, I think lower reward food likely reduces the setpoint itself. Also, higher protein food, restorative sleep, and stress management may help lower the setpoint. But I don’t want to give you the wrong impression here: I don’t have a magic switch for turning off the starvation response completely. These are some things that may dampen it though.
Stephan, the effectiveness of gastric bypass is discussed briefly in your book, but not the suppression of ghrelin which results from the surgery.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5214997/
While gastric bypass is a more drastic step than most people will take to lose and maintain weight, ghrelin production is reduced by eliminating part of the stomach. The following link argues that this ghrelin suppression is effective to reduce hunger.
http://www.obesitycoverage.com/gastric-bypass-vs-gastric-sleeve-surgery/
Hi thhq,
Thanks for reading my book, I hope you enjoyed it. The ghrelin explanation for the effectiveness of gastric bypass was suggested by early evidence but has not been well supported overall and is no longer a leading explanation. Ghrelin is important for appetite control but it doesn’t consistently decline following bypass, and studies in rodent models of bypass (which replicate effects seen in humans quite well) suggest that it isn’t a major player.
But what about the role of ghrelin, apart from bypass? Is hunger a significant factor for weight control? The graph in the following link suggests that it is, in two ways
-Ghrelin is modestly suppressed during intense exercise.
-Ghrelin is strongly promoted by food restriction.
http://www.exercisemed.org/research-blog/ghrelin-the-hormone-that.html
This suggests that for equal calorie deficits exercise would be more effective for weight loss than food restriction.
Once again I’m using your blog as a muse, so please delete if I’m rambling too much…
I’m trying to sort out the reward effect of food – the appetizing or “amuse bouche” effect – from the hunger effect. The following link was interesting.
http://arbl.cvmbs.colostate.edu/hbooks/pathphys/endocrine/gi/ghrelin.html
According to the author obesity suppresses ghrelin. For an obese person hunger isn’t as important as avoiding attractive foods which sustain the obesity. However as weight is lost ghrelin levels rise, and the dieter starts to eat more in reaction to true hunger signaling from ghrelin rather than a food being rewarding. Since the dieter is used to a comfortable stomach rather than a cramped growling stomach, the dieting becomes more and more of a chore. And the problem doesn’t go away once the weight is lost. The weight-reduced person will always have memories of the pleasant obese state instead of the nagging ghrelin signaling for feeding.
Ghrelin isn’t a major player in gastric bypass, but it still is in gastric sleeve right? This suppression is also causing gastric sleeve patients to have a lot of success. My bariatric physician told me that she only really recommends the sleeve now.
https://bariatricjournal.com/gastric-sleeve/
Continuing the musing….bitter flavors have been found effective to suppress ghrelin in both mice and humans.
http://www.nature.com/articles/srep15985
Bitterness is an indicator of toxicity, and triggers the repression of hunger. If I understand correctly, the response to bitterness is a short increase in appetite followed by greatly slowed gastric emptying.
This suggests some N=1 work using two readily available bitter materials: chewing a low dose aspirin tablet before dinner and eating 10g of high cacao chocolate chips around midnight. Even the smell of chocolate has been shown to repress ghrelin
https://www.ncbi.nlm.nih.gov/pubmed/20102728
but I’d prefer to eat the chips rather than sniff them.
The hoped-for result is reduced hunger between 3 and 5 AM. Usually these N=1’s lead nowhere. But hope springs eternal.
On the first try I slept deeply from midnight to 5AM, but at that point the alarm clock in the pit of my stomach went off. Some progress anyway. And Guittard chips taste good at midnight.
The bittersweet chocolate continues to damp AM hunger, but after the first night it has had a non-drowsy effect like a cup of coffee. I’ll try switching to aspirin or quinine.
Not pharmaceutical quinine – just a 3 oz shot of tonic water, which is about 10mg dose, and tonic has never bothered me as a beverage. Much more bitter than chocolate.
Continuing the muse, bitter taste for ghrelin regulation might be most effective ahead of meals, in order to suppress hunger for 3 hours after a meal and reduce snacking. The best source of bitterness appears to be DB, denatonium benzoate, and it works better for women than men.
http://ajcn.nutrition.org/content/early/2017/02/01/ajcn.116.138297
I found some DB, formulated an anti-nail biting preparation. It definitely deterred me chewing my nails. It has a bitterness similar to quinine in tonic water on the back of the tongue, but not mediated by any sweetener. I’ve also tried Swedish Bitters, which have a more vegetal flavor.
I’m backing into bitters as digestive tools, with only general knowledge of aperetifs, digestifs and the trou normand. But not every bitter substance is effective. Tonic and DB are known to be.
A week into this, I’m drinking a shot glass mixture of tonic, Swedish bitters and dissolved DB before every meal. This has a strong and lingering bitter flavor. Ive had no weight loss or gain, but no reduction in eating was planned. Maybe a little less hungry post prandial. Sleep is about the same as before over several nights. If the bitterness effect is reduced gastric for 3 hours after a meal I guess I wouldn’t expect much at 3 AM, 9 hours after a meal. An empty stomach can’t get any emptier.
2 weeks on, the Swedish bitters are gone, leaving a mix of DB and tonic. No weight change, no change in 3 AM hunger, so null effect as a weight loss or sleep tool. The bitterness clears my palate and whets my appetite. There’s a little better sense of fullness after a meal, but that effect is completely gone within 3 hours. I have been reminded of how much I like the bitter flavor of tonic. I recommend Fever Tree for quality but Schweppes is the good inexpensive go-to for N=1 experiments.
Just about to order your book, but in the mean time…Have you any thoughts as to whether increasing muscle mass has an impact on set point? Clearly it helps increase resting metabolism but does a person who, for example, weighs 150lbs have the same set point independent of whether s/he manipulates body composition yet retains same body weight?
Hi Nancy,
I don’t know whether increasing muscle mass impacts the set point, but I do think exercise can influence it. Hope you enjoy the book!
Hey Stephan,
I have just found your research and it is excellent to read. I am interested to know your opinion of individuals with a ‘strict’ set-point. For example, I have a BMI of 27.5 which I can maintain pretty effortlessly eating a diet of 85% whole foods with moderate exercise. Interestingly, the only time I have ever attempted to lose weight I found out that I stop menstruating when I hit a BMI of around 25.5 (I did drop my calories drastically very suddenly but I’m not sure whether that would have contributed to the problem or not). Have you ever found any differences (even anecdotally) between males and females regarding the brain’s response to calorie restriction? And, could someone like me ever break through the 25.5 mark hah.
Thanks 🙂
Hi Francesca,
Some peoples’ weight is regulated more tightly than others, but I’m not aware that it differs between men and women per se. Loss of menstruation is a typical part of the brain’s “starvation response” to fat loss. I believe there are ways to make the brain more comfortable at a lower weight and attenuate the starvation response. It’s too much info to get into here but I discuss them in my book The Hungry Brain.
Thank you for replying. I find it all fascinating so I’ll definitely get round to reading your book.
Hi Stephen,
what are your thoughts on this article which implies that weight regain is due to a lowering of energy expenditure after weight loss rather than an increase in appetite?
thanks,
oops, forgot link: http://weightology.net/weightologyweekly/index.php/free-content/free-content/volume-1-issue-10-insulin-physical-activity-and-weight-regain/physical-activity-and-weight-regain/
So, According to you what is the main reason we tend to loose weight at early stages when we are new to workout or any physical activity and gradually some kind of plateau is achieved and we stop loosing weight at same rate as earlier
“In other words, find ways to keep the brain happy at a lower calorie intake rather than using willpower to continually struggle against the biological drive to eat.”
That about sums it all up Doc. I just stumbled across your site today as today was my 9th day without fast digesting carbs (read: sugar) and i finally gave in and had a bucket of ice cream. 9 days is my max and it almost makes me sick that i cant go on without Carbs (fast digesting) for more than 10 days even.
Is there any remedy for people who are addicted to Carbs? Or will i keep repeating this cycle? Its clear to me that its not a question of will power anymore. I Just need to find a way to keep my brain happy with a lower cal intake (carbs included).
Thank you!
Carbs aren’t “addictive”. You can lose weight on a low carb diet, a high carb diet, or an in-between carb diet. You need to find a diet that you can comfortably stick with. It needs to be high-satiety, at a level of restriction that does not leave you feeling so hungry or so deprived that you abandon the diet in 9 days, and it needs to be a diet that allows you to transition into long-term maintenance. You may need to experiment a little to find something that works for you. You also need to work on the mental aspects of overeating; food-thought triggering activities, food for stress and fatigue relief, food as a hobby, etc. It’s not easy, but people do succeed. However, worrying about macronutrients is undoubtedly one of the least important elements of successful weight loss.
If that can be of any help, there’s this dude who’s on an ice-cream diet.
http://www.menshealth.com/weight-loss/ice-cream-diet
I don’t recommend this “diet”, I am just making a point that in the end, calories are what matters most. So yes, it is important to find a diet that you feel comfortable with and prevents you from overeating all the time.
Love your work Dr. Guyenet! Thanks for so much free high quality professional insight on your websites. Your book is honestly one of the best and should be required reading before any other diet book is read and seen through it’s principles of obesity.
I’ve tried the low food reward of natural whole foods approach while also controlling my home environment. It works until the starvation response kicks in. Well, I should say it would continue to work if the home good environment was the only foods I ate. Knowing the bad high food reward food is only a quick drive away, the starvation response wins again darn it.
I’m wondering if a 80-90% good and a 10% planned, limited amount of high food reward daily would allow keeping the brain happy at the lower level. Thoughts on such an approach? Or do you recommend an all pure low reward approach, or are planned cheats just playing with fire and doomed to fail or is it all just individualized and experiment and see what works for you to keep the brain happy at lower levels? Planned cheats seem like the opposite of “dampening” the appetite but still a “managing” approach is why I ask. Thanks.
I like Weight Watchers for just this reason. By keeping track of my intake, I know if I can afford a little gelato or not. Some people find tracking intake annoying, but after I realized that it costs me less time daily than toothbrushing, I recognized that it is actually really freeing. WW also divides their silly “points” into a daily and weekly pool and that also helps me manage what I eat without feeling deprived or crazy because I overshot my daily goal.
I’ve been counting calories for 10 years. I started with carb counting to control diabetes using exchanges
http://glycemic.com/DiabeticExchange/The%20Diabetic%20Exchange%20List.pdf
After 3 months of this I lost weight and my diabetes symptoms went away. I kept counting, but switched from counting carbs to calories. I still count exchanges but now use units of 70 calories, which is very similar to Weight Watcher points. The mnemonic of points is easier to keep in my memory than running calorie tallies. Unlike other diet systems counting allows me to eat anything I want so long as I don’t exceed my daily quota of points. I also count exercise using the same method, and at the end of the day I have a tally of CICO points which I track in a diary. [Calories eaten – (calories exercise + BMR)] needs be close to zero or I start to gain weight.
Hello!
I want to point out an underappreciated side-effect of a low-reward diet. It has its own different kind of reward.
I can easily eat a whole pizza and feel “full” in the sense that I couldn’t eat any more, but I don’t feel satisfied. But when I’m on a low-reward diet, a meal with protein and fiber gives me a feeling of physical satisfaction that I never experience when I’m eating junk. It’s a very comfortable sensation of a full belly which gives a very pleasant, cosy feeling. It’s like the feeling of curling up with a warm soft blanket. I imagine it reduces stress since it feels so relaxing. When I’m on a poor diet I feel stressed after I eat a meal, not just from guilt, but from the lack of satisfaction.
The only problem is that it takes a few days of not eating any high-reward foods before this kicks in, so it takes some patience.
I think this is worth pointing out because I find it very motivational. When you think of a low-reward diet, you imagine it will be misery, but it actually gives you another kind of physical pleasure. Perhaps obese people on high-reward diets keep eating in frustration as they are subconsciously trying to get this satisfying feeling but they are never able to.
Thanks, Layla, for the excellent description of the comfort factor of a low-reward diet. I know exactly what you are talking about, low-reward food has the same effect on me, and that’s why I prefer to eat it. I think you may be on to something regarding the different kind of physical pleasure deriving from a type of satisfaction that obese people subconsciously seek – but fail to find – in their high-reward diets.