This is a list of references I am likely to cite in my debate with Gary Taubes on the Joe Rogan Experience on March 19. During the debate, I’ll be calling out numbers corresponding to the references below. Scroll down to the number I mentioned to see the references for yourself.
Gary and I prepared a document explaining our models that you can download here. For context, I am favorable toward low-carbohydrate diets, but the references below mostly reflect issues that Gary and I are likely to disagree on.
The brain
1. The brain regulates body fatness
This is supported by a mountain of evidence going back 180 years. The brain, and particularly a non-conscious part of the brain called the hypothalamus, contains the only known system in the body that regulates body fatness. Damage to specific parts of the hypothalamus cause severe obesity or leanness in animals and humans, and experimentally controlling the activity of specific groups of neurons alters body fatness in animal models. See these review papers for an overview of some of what we know. I also give a lay-friendly overview of this research in my book The Hungry Brain.
Nearly all anti-obesity drugs that are currently approved by the FDA target the brain, including liraglutide, lorcaserin, phentermine/topiramate, and naltrexone/bupropion. The only exception is orlistat, which reduces dietary fat absorption in the gut. Interestingly, one of the most effective of these weight loss drugs, liraglutide, increases post-meal insulin secretion.
2. The genetics of obesity point to the brain, not fat cells or insulin, as the primary determinant of body fatness
Obesity risk is strongly influenced by genetics, and recent studies have begun to uncover the specific genetic differences that make some people fatter than others. This gives us the opportunity to see what these genes do, providing a window into the causes of common obesity. Results show that gene variants that impact body fatness are primarily related to brain function— not primarily insulin or fat cells (although many mechanisms may contribute to a smaller degree). Importantly, these (GWA) studies are unbiased because they examine the whole genome without preconceived notions about which genes or biological pathways might be important.
The largest and most recent study on the genetics of body fatness concluded that “[body mass index]-associated genes are mostly enriched among genes involved in neurogenesis and more generally involved in the development of the central nervous system.”
3. The brain regulates appetite
This should be obvious, because appetite is a motivational state that is generated by the brain– like all motivational states. In fact, we know so much about how appetite-regulating brain circuits work that researchers can turn appetite on and off at will by controlling specific types of neurons in animals. Here are a few review papers that give a broad overview of much of what we know. I give a lay-friendly overview of this research in my book The Hungry Brain.
4. The determinants of appetite are complex and cannot be reduced to glucose and insulin
The brain determines hunger and satiety by in response to signals it receives from inside and outside the body. From inside the body, the brain receives information about the body’s energy status. This comes from neural signals and hormones ascending from the small intestine, pancreas, liver, and fat tissue that tell the brain how much food is in the gut, what type of food it is, and how much fat the body contains.
From outside the body, the brain receives signals about what food cues surround you, and this can also stimulate hunger (think about smelling pizza). All of this information is integrated by the brain, which uses it to generate the overall feeling of hunger or satiety. For a review of some of the research on this see this paper.
5. Lean people, and people with obesity, both “defend” their current level of body fatness against fat loss
When people lose fat, their brains initiate a sort of “starvation response” that favors the regain of lost fat over time. This is familiar to most people who have dieted: increased hunger, more cravings, reduced metabolic rate, and eventual weight regain. This is the brain’s response to fat loss, regardless of whether a person starts off lean or with obesity. This suggests that the “setpoint” around which body fatness is regulated is increased in obesity, since the brain nonconsciously “defends” the obese state. This is one of the key reasons why weight loss is challenging and often temporary.
We know this effect is due to a decline in the fat-regulating hormone leptin during weight loss, because replacing leptin to the pre-weight-loss level largely prevents this “starvation response”. Therefore, the reason people with obesity “defend” a higher level of fat mass is that their brains require a higher level of leptin to feel like they aren’t starving.
6. Single-gene mutations that lead to obesity in humans act in the brain
A number of single-gene mutations have been identified that lead to severe obesity in humans and animals. These are all related to the brain systems that regulate body fatness, and particularly the fat-regulating hormone leptin. I’m not aware of any mutations in insulin or fat cell-related genes that cause obesity.
Calories
7. People with obesity eat, and expend, more calories than lean people
Studies using accurate methods consistently show that people with obesity eat, and expend, about 20-35 percent more calories than lean people, after correcting for height, sex, and physical activity level. Calorie intake and expenditure increase continuously in parallel with body weight. Since calorie intake is equal to calorie expenditure in a weight-stable person, measuring calorie expenditure is the simplest and most accurate way to demonstrate that intake and expenditure are both elevated.
8. Reducing the calorie intake of a person with obesity to that of a lean person causes weight loss
People with obesity consume about 20-35 percent more calories than lean people. Tightly controlled studies have demonstrated that restricting calorie intake by about this much in people with obesity causes rapid weight and fat loss, demonstrating that the elevated calorie intake is required to sustain the obese state.
9. Calorie intake, not carbohydrate intake, is the main dietary determinant of body fat loss
Several controlled feeding studies have compared the effect of calorie-restricted diets with different proportions of fat and carbohydrate on weight and fat loss. Fat loss is determined primarily by the number of calories in the diets, not the proportion of fat or carbohydrate, even when insulin levels differ substantially. This was confirmed by a recent meta-analysis (study of studies) of 20 studies, which concluded that “for all practical purposes ‘a calorie is a calorie’ when it comes to body fat”.
10. Changes in body fatness as a result of diet depend primarily on calorie intake, not carbohydrate intake
This was demonstrated by a meta-analysis (study of studies) of 20 tightly controlled studies in which people were fed diets that differed in carbohydrate and fat content, but not in protein or calorie content. In other words, same calories, different carb:fat ratio. Calorie-for-calorie, higher-carbohydrate diets led to slightly more body fat loss than higher-fat diets, but the difference was very small. The researchers concluded that “for all practical purposes ‘a calorie is a calorie’ when it comes to body fat.”
11. Energy expenditure (metabolic rate) is scarcely affected by differences in carbohydrate and fat intake
This was demonstrated by a meta-analysis (study of studies) of 28 tightly controlled feeding studies. Altering the proportion of carbohydrate to fat in the diet had very little impact on metabolic rate when calorie and protein intake were the same, although metabolic rate was slightly higher on higher-carbohydrate diets.
A recent study received a lot of attention for finding that a very-low-carbohydrate diet led to a higher metabolic rate (+209 kcal/day) than a low-fat diet in people maintaining weight loss. While this study was rigorous and innovative in many ways, some of the data it collected are clearly erroneous because they appear to break the first law of thermodynamics, which is physically impossible. Once the erroneous data are removed, the effect size shrinks, loses its statistical significance, and becomes similar to the other 28 studies that were conducted before it.
Carbohydrate, sugar, and fat
12. Low-fat, high-carbohydrate diets cause weight loss, even without deliberate portion control
The most rigorous study on this is the DIETFITS randomized controlled trial, which was partially funded by Gary Taubes’s organization NuSI. In 609 people, a whole-food low-fat diet resulted in 12 pounds (5.3 kg) of weight loss at one year, not significantly different from the 13 pounds (6.0 kg) of weight lost on a whole-food low-carbohydrate diet. Neither diet included advice to restrict calorie intake. The low-fat diet supplied about twice as much carbohydrate, and 1.5 times as much sugar, as the low-carbohydrate diet, although neither diet was high in sugar. It also had about twice the glycemic load.
Another study conducted at the University of Washington tested the effect of dietary fat restriction on calorie intake and body weight in 16 overweight people using very careful measurements. When they were transitioned to a low-fat diet (15%) and told to eat as much as they wanted, their daily calorie intake dropped by 291 calories and they steadily lost weight and body fat over a 12-week period, losing 8 lbs (3.8 kg) total. Importantly, their total carbohydrate intake increased from 253 grams per day to 318 grams per day. Here’s the graph showing daily calorie intake and weight change (circles = calorie intake, diamonds = weight):
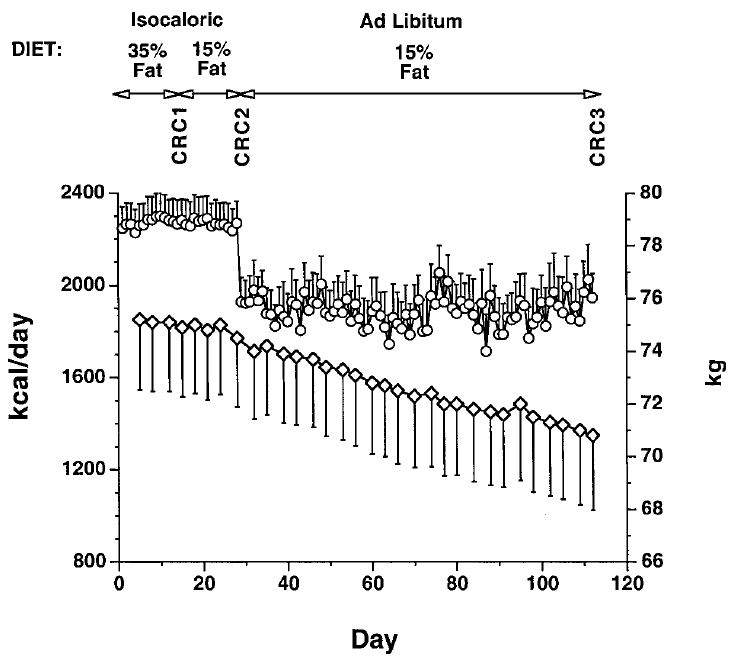
A meta-analysis (study of studies) representing 16 low-fat diet studies also reported that low-fat diets without deliberate portion control cause weight loss.
13. A low-calorie diet composed almost entirely of refined carbohydrate, including sugar, causes substantial weight loss
This paper discusses 106 patients who lost an average of 140 pounds (64 kg) and improved their blood sugar control eating the “rice diet”, a diet composed almost entirely of white rice, fruit, juice, and white sugar. Here is a photo of one man before and after the diet, from the paper:
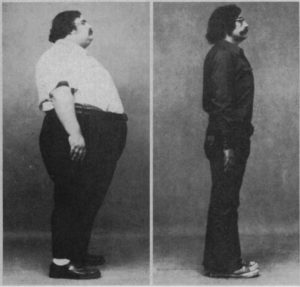
The weight loss version of the rice diet was low in calories, supplied over 200 grams of mostly refined carbohydrate per day, and patients also increased physical activity. This demonstrates that refined carbohydrate does not override the impact of calorie intake and physical activity on body fatness.
The rice diet was also effective for treating diabetes, whether or not it was calorie-restricted.
14. Dietary fat can be fattening in a variety of nonhuman species
A review paper on this topic states that “With few exceptions, obesity is induced by high-fat diets in monkeys, dogs, pigs, hamsters, squirrels, rats, and mice.” There are hundreds of studies demonstrating this, but the most conclusive one was published in 2018 by John Speakman’s lab. They fed five different strains of mice diets varying in fat, carbohydrate, and protein content and showed that body fatness was primarily linked to the amount of fat the mice were eating. Body fatness increased as dietary fat increased up to 60% of calories, and then body fatness declined as dietary fat increased further (this is consistent with other research showing that the very-high-fat, very-low-carb ketogenic diet promotes leanness in mice, as it does in humans). Diets high in sugar and refined starch were not particularly fattening. The takeaway is that the most fattening diet contains abundant fat and carbohydrate together, while diets at the extremes (very-low-fat and very-low-carb) are more slimming.
15. Dietary fat can be fattening in humans
This really depends on context, since low-carbohydrate, high-fat diets tend to be slimming relative to diets that are high in both carbohydrate and fat. Nevertheless, unrestricted diets rich in fat tend to lead to higher calorie intake and fat gain relative to unrestricted diets rich in carbohydrate. This seems to be related to the high calorie density and palatability of fat.
Overfeeding fat under controlled conditions also causes body fat gain.
16. Fat and carbohydrate are equally fattening when overconsumed
Two randomized controlled trials have reported the effect of overfeeding the same number of calories of high-carbohydrate vs. high-fat diets in humans, and both reported equal fat gain.
- Horton and colleagues increased the calorie intake of volunteers by 50 percent by adding only fat vs. only carbohydrate for 14 days under tightly controlled conditions, and measured changes in body fat mass using gold-standard hydrostatic weighing. After 14 days, both groups had gained 3.3 lbs of fat, despite higher insulin levels in the high-carbohydrate group. In the high-carbohydrate arm, most of the extra carbohydrate was refined starch and sugar. In the high-fat arm, most of the extra fat was dairy fat (cream, butter).
- Lammert and colleagues increased the calorie intake of volunteers by 1,194 calories per day for 21 days under tightly controlled conditions. Subjects were fed either a high-carbohydrate (78% CHO, 11% fat) or a high-fat (31% CHO, 58% fat) diet containing the same amount of calories and protein, and the researchers measured changes in body fat mass using gold-standard hydrostatic weighing. After 21 days, both groups had gained 1.8-1.9 lbs of fat.
17. Sugar intake has been declining for 20 years in the US and 50 years in the UK, while obesity and diabetes rates have risen
Here is a graph showing sugar intake and the obesity rate in the US between 1980 and 2013. The sugar data include all added sugars such as table sugar, honey, and high-fructose corn syrup, but not sugars naturally occurring in fruits and vegetables.
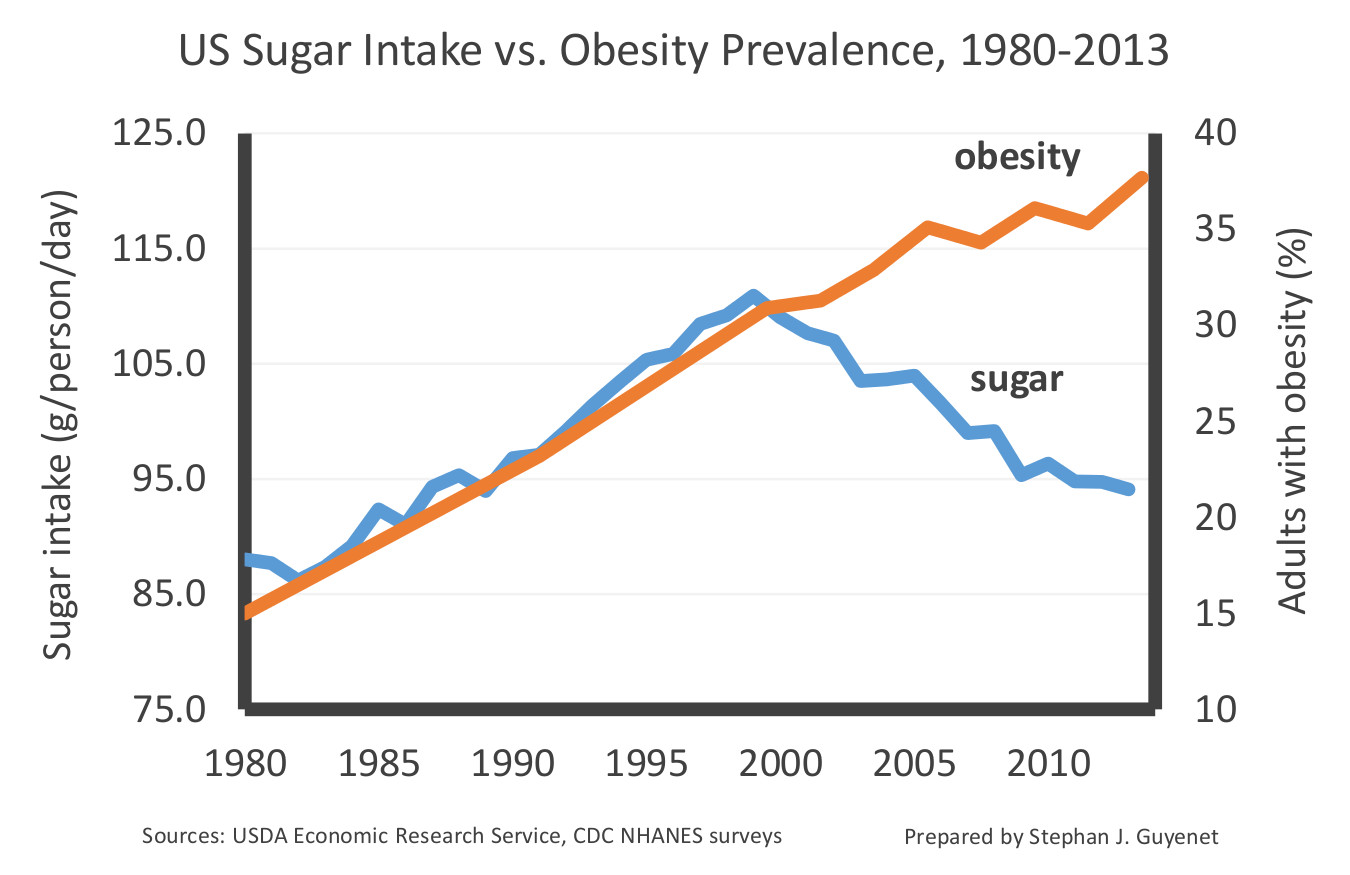
This graph illustrates the lack of correlation between sugar intake and obesity rates after 1999. This decline in sugar intake has been confirmed by several independent lines of evidence. The data suggest that sugar intake has dropped by 15-23 percent over the last two decades, primarily because people are drinking fewer sweetened beverages. Diabetes rates have also increased during the decline in sugar intake.
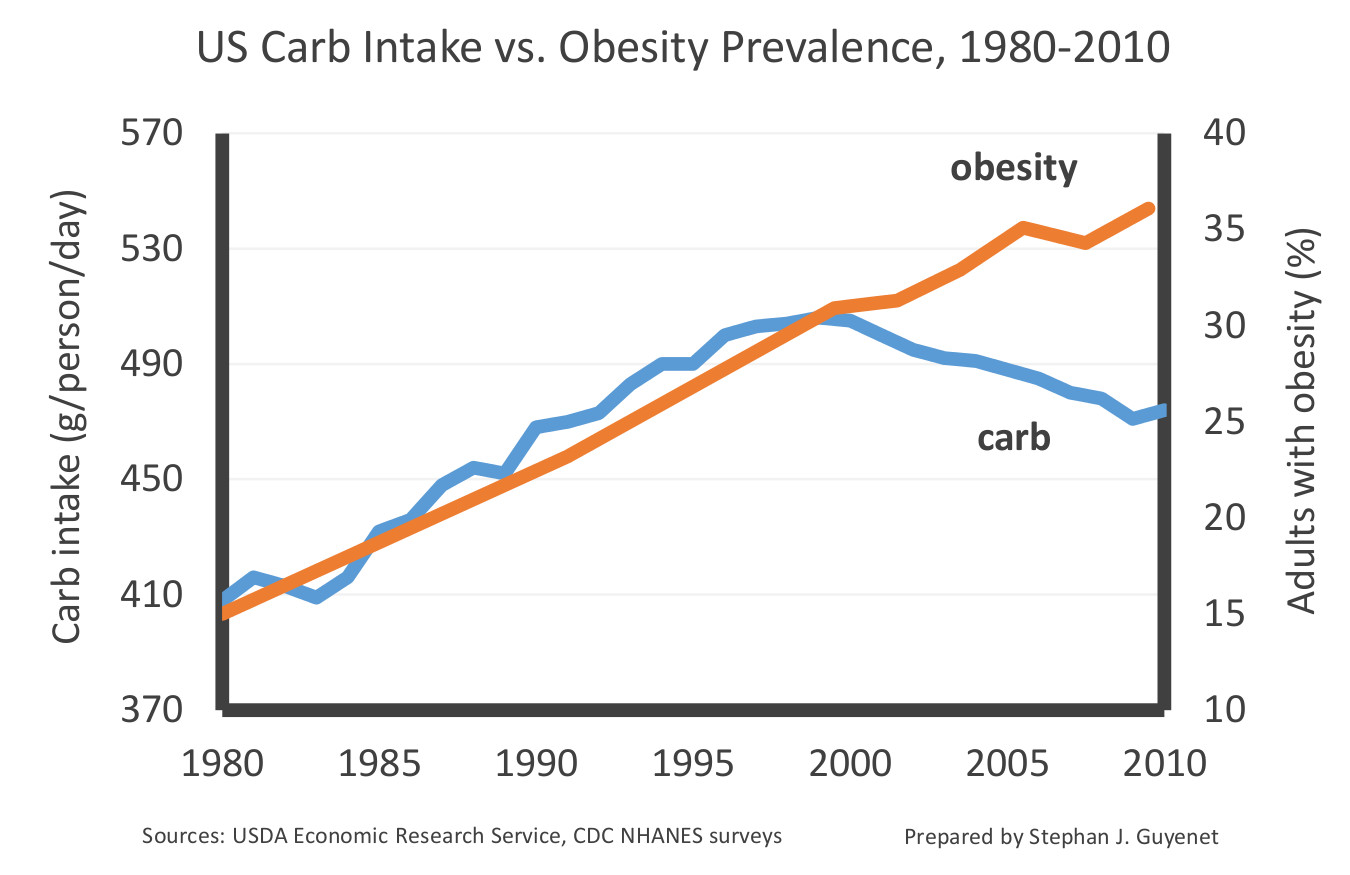
Overall carbohydrate intake has also declined over the last two decades in the US:
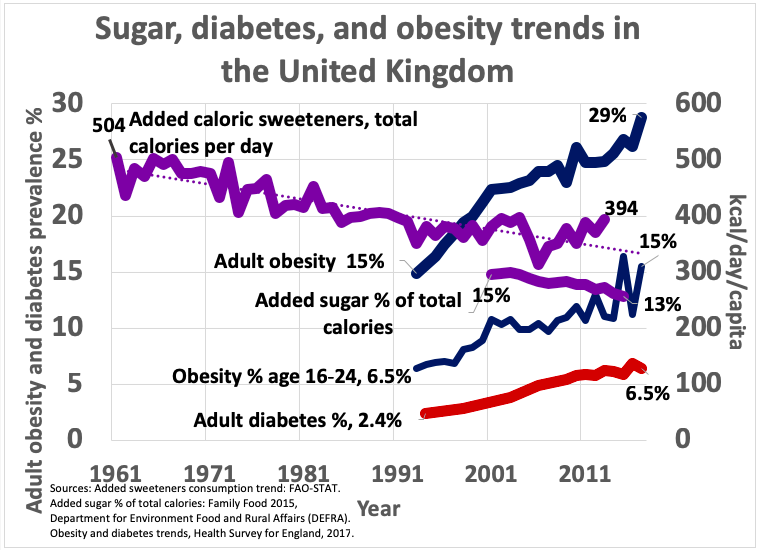
This graph shows that sugar intake has declined by 22 percent in the UK over the last 50 years, while obesity and diabetes rates have increased substantially (prepared by Kevin Bass):
18. Carbohydrate, fat, and protein intake in the US over the last century
This graph represents absolute carbohydrate, fat, and protein intake in the US since 1909. It’s based on US Department of Agriculture data.
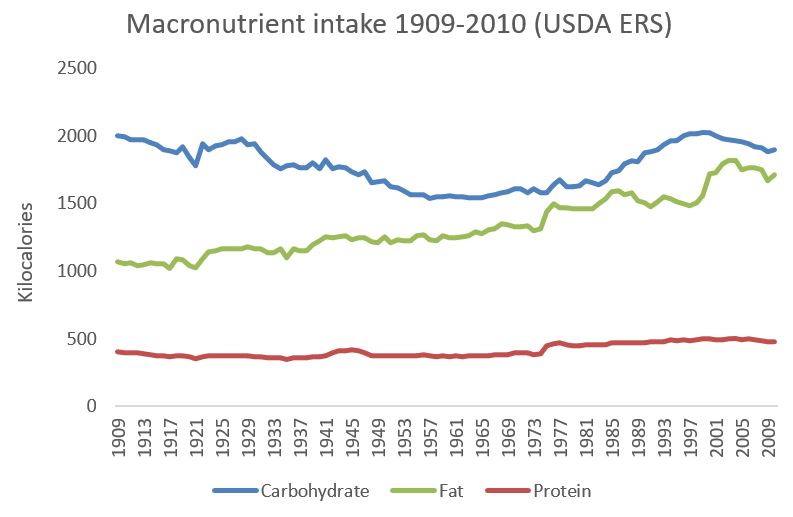
Note that fat is the only macronutrient that has consistently and substantially increased over time. Carbohydrate intake declined in the 1940s-50s, rose again in the 1980s-90s, and has been declining since 1999.
Here are the same data, represented as percentages of total calorie intake:
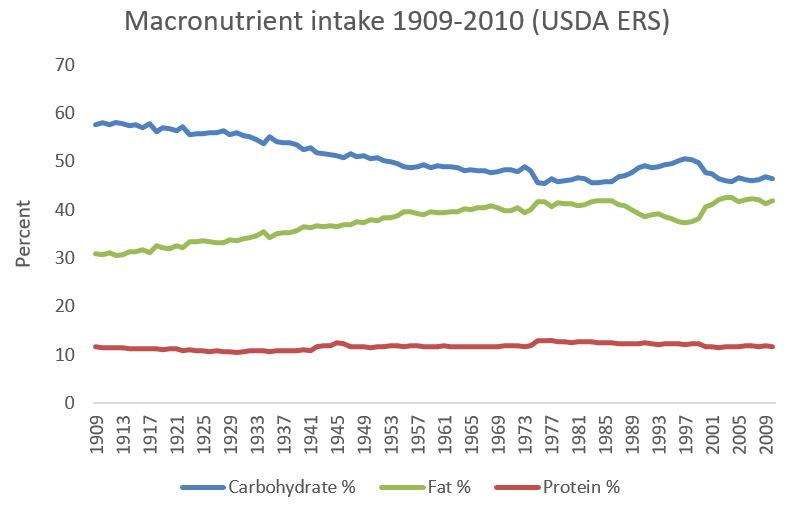
Aside from a little bump in the 1980s and 1990s, as a percentage of calories the US diet has progressively become higher in fat and lower in carbohydrate.
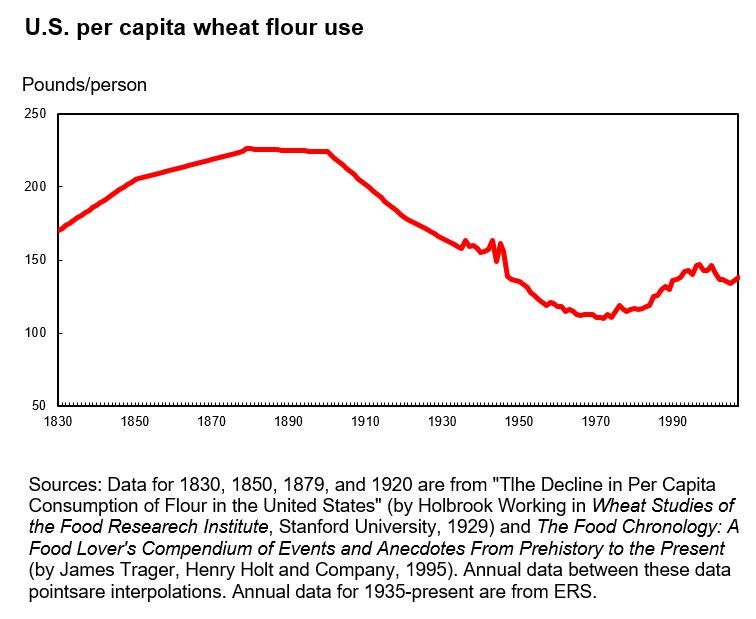
19. White flour intake was much higher in 1900 in the US than it is today
I downloaded this graph directly from the US Department of Agriculture website, although the web page I downloaded it from is no longer online. It speaks for itself. The large majority of this flour would have been refined, white flour from the late 1800s onward.
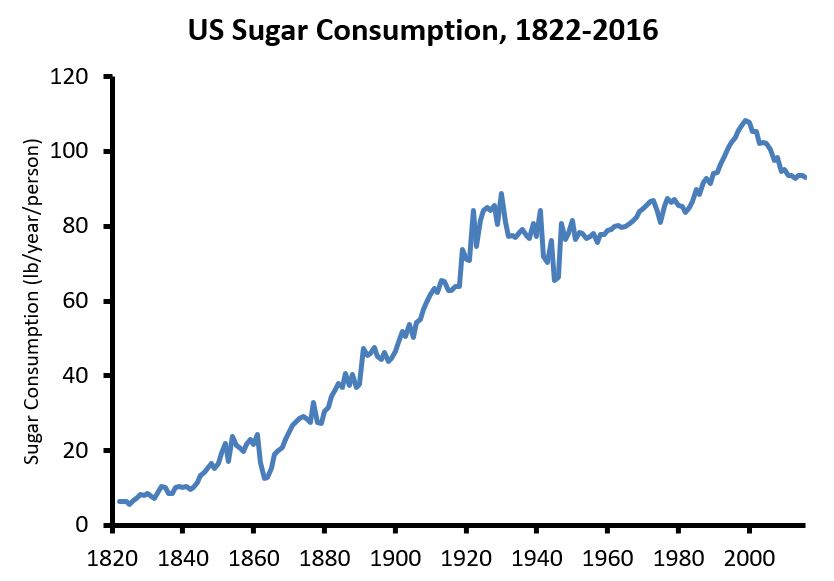
20. Sugar intake in the US, 1822-2016
This graph represents added sugar intake, including honey and high-fructose corn syrup but not fruit sugars, from several data sources:
21. Cultures with high intakes of sugar but otherwise healthy diets and lifestyles do not have obesity or diabetes
A well-studied Tanzanian hunter-gatherer population called the Hadza gets 15 percent of its average year-round calorie intake from honey, plus fruit sugar on top of it. This approximates US sugar intake, yet the Hadza do not exhibit obesity, diabetes, or cardiovascular disease.
Another mostly hunter-gatherer population, the Mbuti pygmies of the Congo, also ate large amounts of honey at the time they were studied:
The Mbuti of the Congo forests eat large amounts of honey. According to Ichikawa (1981), honey is their favorite food. At times, during the rainy season up to 80% of the calories in their diet come from honey.
They did not exhibit obesity or insulin resistance.
The Kuna of Panama are interesting because they generally have a non-industrialized diet and lifestyle, except that they obtain white sugar and a few sugar-containing foods via trade. Their diet is 65 percent carbohydrate and 17 percent sugar (95 g/d), according to calculations I did based on dietary intake data. That is more sugar than the average American currently consumes, although 62 percent of their sugar intake is from fruit. The Kuna are lean and have excellent cardiovascular health.
This does not exonerate sugar by any means, but it does suggest that it is unlikely to be the primary cause of obesity, metabolic disease, and cardiovascular disease.
22. During the Cuban economic crisis, sugar intake rose, calorie intake declined, and obesity plummeted
From 1989 through about 1995, the Cuban economy collapsed. Calorie intake declined, fat intake declined, and the diet became very rich in sugar and other refined carbohydrates:
By 1993, carbohydrate, fat, and protein contributed 77 percent, 13 percent, and 10 percent of total energy, respectively, whereas in 1980 their respective contributions were 65 percent, 20 percent, and 15 percent (8). The primary sources of energy during the crisis were sugar cane and rice.
Sugar intake “rose to 28% of total energy intake“. A shortage of gasoline meant that people began walking and riding bicycles more, such that total physical activity increased. During this time, the prevalence of obesity decreased by about half, then eventually rebounded as the crisis resolved and the diet went back to normal. The prevalence of underweight only increased slightly during the crisis, indicating that the decline in obesity was not due to widespread starvation.
23. Cultures with very high intakes of carbohydrate tend to be lean, even if the carbohydrate is white rice
The traditional diet of most agricultural societies around the world is high in carbohydrate and low in fat. Many of these cultures have been studied, and they are typically lean with low rates of diabetes and cardiovascular disease, even if they have abundant food. The book Western Diseases: Their Emergence and Prevention contains many such examples.
In 1990, Staffan Lindeberg, MD, PhD conducted a detailed survey of the diet, lifestyle, and health of the residents of Kitava, a Melanesian island scarcely touched by industrialization. He found a diet based on starchy plant foods (African yam, sweet potato, taro, cassava), fruit, vegetables, seafood, and coconut, with 69 percent of calories coming from carbohydrate (compared to ~49% in the US currently). Despite food abundance, none of the 247 Kitavans Lindeberg examined had obesity or diabetes, even in middle age and older. They had no sign of cardiovascular disease and were unfamiliar with its symptoms. Here is a photo of a Kitavan man being examined by Lindeberg (courtesy of Staffan Lindeberg):

The book Western Diseases: Their Emergence and Prevention contains historical data on the Japanese diet between 1950 and 1975, a time during which obesity was even more rare in Japan than it is today (p. 338). In 1975, long after postwar food shortages were over, the Japanese diet was 62 percent carbohydrate, mostly from white rice. The Japanese ate it daily as their primary staple food, and still do, yet the prevalence of overweight and obesity were, and continue to be, lower than any other industrialized nation. People who emigrate from Japan to the US and begin eating a diet lower in white rice and higher in fat become much heavier, demonstrating that their leanness is not just genetic.
Cross-country comparisons show that nations with carbohydrate-focused diets tend to have the lowest obesity rates, while nations that eat a similar amount of carbohydrate and fat have the highest obesity rates (reproduced from this post with permission from Cian Foley):
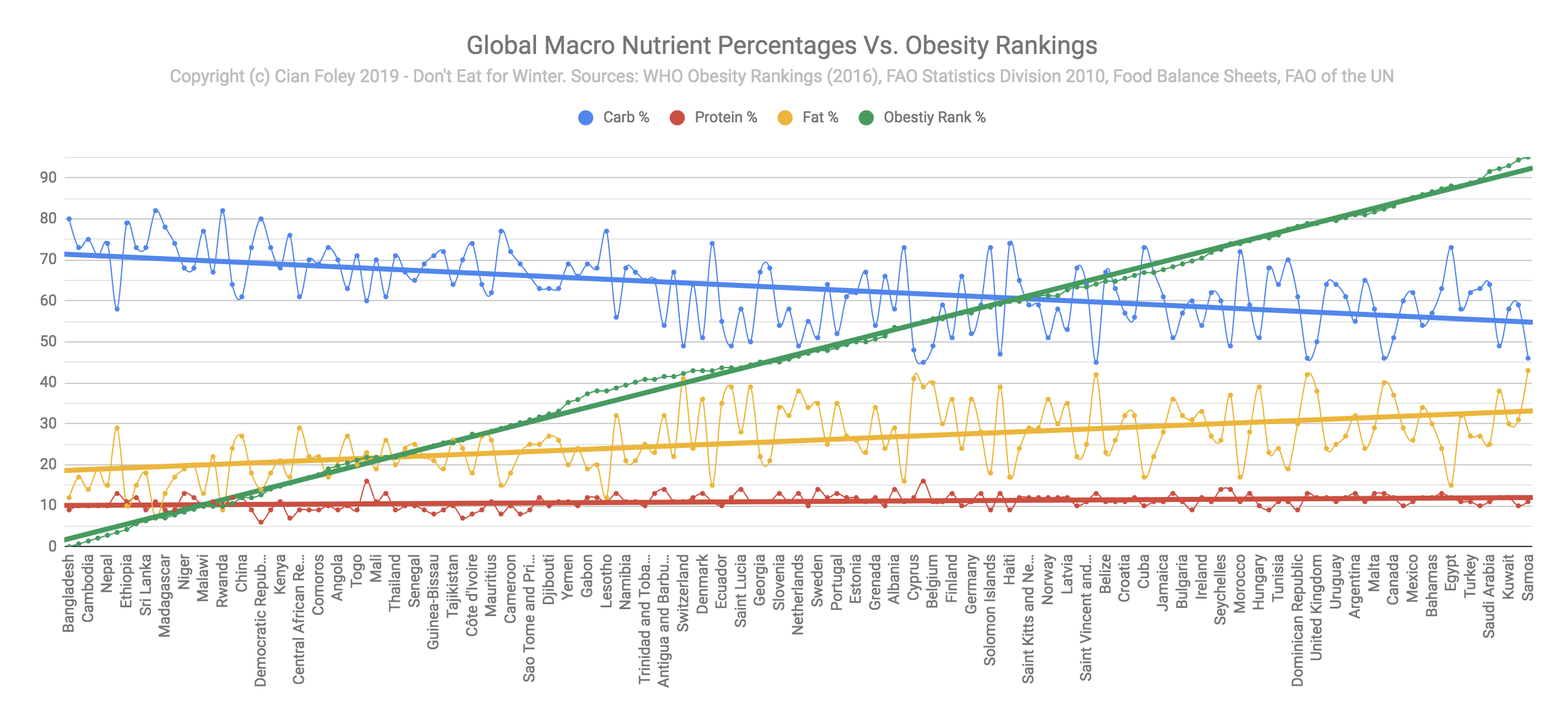
There are certainly explanations for this besides macronutrients– such as poverty– but at the very least this shows that carbohydrate is not an overriding driver of fat gain.
24. In human randomized controlled trials, sugar is only fattening by virtue of its calories
A meta-analysis of 30 randomized controlled trials reported that increasing sugar intake does not increase body weight unless total calorie intake increases. Importantly, calorie intake does often increase when people increase their consumption of sugar-sweetened beverages, so sugar-sweetened beverages aren’t off the hook.
Fructose is half of table sugar (sucrose). A meta-analysis of 31 fructose feeding trials concluded that “Fructose does not seem to cause weight gain when it is substituted for other carbohydrates in diets providing similar calories. Free fructose at high doses that provided excess calories modestly increased body weight, an effect that may be due to the extra calories rather than the fructose.”
25. In randomized controlled trials, low-carbohydrate and low-fat diets yield similar (unimpressive) long-term weight loss results
In real-life conditions, low-fat and low-carbohydrate diets both produce modest weight loss in the average person, usually less than 17 pounds at the 12 month mark. Most meta-analyses (studies of studies) report that weight loss differences between the two diets are “minimal” at 12 months, although low-carbohydrate diets usually fare better up to 6 months. The differences in weight loss between individuals on the same diet are much larger than the differences between diets.
People often dismiss these findings because the diets weren’t lower in carbohydrate. This is a valid critique but it also applies to the low-fat diets used in these studies, which were typically not very low in fat. Also, the fact that people usually don’t restrict carbohydrate or fat as much as instructed shows that most people find the diets hard to sustain in real life.
26. Meta-analyses of ketogenic diet studies
In a quick literature search, I found one meta-analysis of ketogenic diet studies, which reports long-term weight loss after 12 or more months. The ketogenic diets caused 2 pounds (0.9 kg) more weight loss than the low-fat diets.
27. Adherence to ketogenic diets tends to be mediocre
A detailed review of the ketogenic diet literature concluded:
People who are left to their own devices generally do not adhere well to the diet. They typically start strong, but have a hard time adhering to the diet after 1-3 months. Carb intake increases and ketone levels drop.
I have little doubt that better adherence would lead to greater weight loss, but this is true of any weight loss diet.
28. The Virta Health study
The Virta Health study is a non-randomized, controlled trial that reported outcomes for people with type 2 (common) diabetes who followed a ketogenic diet with intensive diet and medical support, vs. care as usual. At one year, participants in the ketogenic diet arm lost 30 pounds (14 kg), lowered their average blood glucose substantially, and greatly reduced their need for diabetes medications other than metformin. Metabolic and cardiovascular risk markers, and the low adverse event rate, suggest that the diet was safe over this period of time. Two-year outcomes suggest that people had begun rebounding back to their original weights and health, but maintenance was still good by the standards of diet interventions.
The Virta Health study reports findings that are quite a bit more impressive than other studies of low-carbohydrate and ketogenic diets. Here are a few reasons why that might be the case:
- Non-randomized trials like the Virta Health study tend to report better results than randomized trials because people self-select into the group they prefer.
- Participants may have been highly motivated because they had type 2 diabetes and the Virta Health program is expensive.
- The Virta Health program provides intensive support to help people adhere to the diet, so adherence was better than in most studies.
29. Eating sugar does not impair weight loss on calorie-reduced diets
I am aware of three weight loss studies that have compared the weight loss response in people eating different amounts of sugar. All three reported that sugar had no impact on weight loss in the context of calorie restriction. One of the studies compared a diet that was 43 percent sugar (sucrose) to one that was 4 percent sugar. Both diets caused the same amount of weight and fat loss over 6 weeks.
Diabetes
30. Weight loss greatly reduces the risk of developing type 2 (common) diabetes
This was conclusively demonstrated by several large, multi-year, randomized controlled trials with actual diabetes diagnoses as the endpoint. One of these trials was the Diabetes Prevention Program study, in which modest weight loss with a calorie-restricted low-fat diet and exercise caused a 58% reduction in the risk of developing diabetes over 2.8 years in 3,234 people with prediabetes. The benefit was largely explained by weight loss, and it was still detectable 15 years later. The diet focused on fat and calorie reduction and was not low in carbohydrate or sugar.
At least four other randomized controlled trials, conducted in four different countries, in people of different races and ethnicities, produced very similar results.
31. Reducing fat intake, without reducing carbohydrate intake, causes weight loss and reduces type 2 (common) diabetes risk
The drug orlistat reduces the absorption of dietary fat in the gut by about 30 percent, without impacting carbohydrate absorption. Large, multi-year placebo-controlled randomized trials have shown that orlistat causes weight loss and reduces the risk of developing type 2 diabetes by 37 percent. This demonstrates that weight loss and protection against diabetes can occur as a result of reducing fat intake, without reducing carbohydrate intake. Orlistat is a FDA-approved weight loss drug.
32. Exercise substantially reduces the risk of developing type 2 (common) diabetes
This was conclusively demonstrated by the Da Qing trial, a 6-year randomized controlled trial. The trial reported that increasing exercise reduces the risk of progressing from prediabetes to diabetes by 46 percent. A second randomized controlled trial reported that two years of supervised aerobic exercise reduced the risk of developing type 2 diabetes by 72 percent, and resistance training reduced the risk by 65 percent. The fact that the exercise was supervised is important– it means the participants actually did it.
33. Weight loss via a temporary very-low-calorie diet durably puts type 2 (common) diabetes into remission
This was conclusively demonstrated by large, a one-year randomized controlled trial in people with type 2 diabetes. The intervention was 3-5 months of a very-low-calorie diet (825–853 kcal/day of a beverage, 59% C, 13% F, 26% P) followed by diet/lifestyle support for the remainder of the year. This caused diabetes remission in 46 percent of people. In other words, 46 percent of people were able to eat normally again and go off medication without experiencing diabetic blood sugar levels for months after the end of the low-calorie diet. Among those who lost the most weight, 86 percent went into remission.
Insulin and insulin resistance
34. Insulin resistance predicts a variety of age-related diseases
In 2001, Gerald Reaven’s group published a very simple but powerful study. They directly measured insulin sensitivity in 147 lean and overweight middle-aged people and waited 6 years to see who got sick, who died, and who didn’t. In the third of the group that was the most insulin resistant, 36 percent developed a health condition or died over the next 6 years, while in the third of the group with the least insulin resistance, none developed a health condition or died. The health conditions that occurred in the insulin-resistant group included high blood pressure, coronary heart disease, stroke, cancer, type 2 diabetes, and death.
35. People with higher insulin levels don’t gain more weight over time than people with lower insulin levels
Many studies have measured insulin levels in an attempt to predict who will gain weight and who will not. Some studies have measured fasting insulin levels, others carbohydrate-stimulated insulin levels. These studies were collected together in a review paper. The research overall suggests that there is no reliable connection between current insulin level and future weight gain. Of the 22 studies included in the review, 5 reported that higher insulin is correlated with more weight gain, 9 reported no correlation between the two, and 8 reported that higher insulin is correlated with less weight gain.
36. Weight loss increases insulin sensitivity, regardless of whether it’s achieved by a low-fat or low-carbohydrate diet
This was demonstrated by a randomized controlled trial in 2009. The low-fat (60% carbohydrate) and low-carbohydrate (20% carbohydrate) diet groups each cut about 500 calories per day and both lost about 9 pounds (4 kg) of body fat over 8 weeks. At the end, insulin sensitivity improved, with no significant difference between groups. Importantly, this study used a gold-standard method for measuring insulin sensitivity, making this result more informative than the vast majority of other low-fat vs. low-carb studies, which tend to indirectly estimate insulin sensitivity using the less reliable HOMA method.
This finding is echoed by an earlier randomized controlled trial in which a 21-day low-fat vs. low-carbohydrate diet, without weight loss, yielded no differences in insulin sensitivity in people with mild type 2 diabetes. This trial also used a gold-standard measure of insulin sensitivity.
37. Insulin resistance is “an appropriate response to nutrient excess“
Exposing cells and tissues to excess energy causes insulin resistance, regardless of whether the excess energy comes from carbohydrate (glucose), fat (fatty acids), or protein (amino acids). This probably happens because excess energy is toxic to cells, so cells turn down their insulin responsiveness in order to take in less energy.
Nutrient excess causes insulin resistance in intact humans as well. Temporarily increasing circulating levels of fat, carbohydrate, or protein in human volunteers markedly impairs their insulin sensitivity.
38. When fat cells “fill up” due to fat gain, energy spills over onto other tissues, causing insulin resistance
As fat tissue expands, fat cells become less effective at taking up fat, especially after meals, and this fat spills over onto other tissues. Depending on underlying genetic factors, different people have different “personal fat thresholds” at which fat gain causes their fat tissue becomes less effective at trapping fat, resulting in insulin resistance and often diabetes.
39. The impact of foods on blood glucose and insulin has little do with how filling they are
This was most clearly demonstrated by a study that fed people the same number of calories from 38 common foods, including bread, potatoes, beans, meat, fruit, peanuts, candy, and pastries, and measured fullness ratings for two hours. Fullness was unrelated to blood glucose responses, and weakly related to insulin responses (higher insulin release correlated with greater fullness).
In contrast, simple food properties like calorie density, protein content, and palatability were strongly related to fullness.
40. Insulin resistance is caused (in large part) by exceeding the unique storage capacity of your own fat tissue
Studies have examined the genetics of insulin resistance, giving us insight into the biological pathways that underlie insulin resistance in the general population. These studies tend to find that gene variants that reduce the storage capacity of fat tissue are associated with insulin resistance.
People with different genetic backgrounds tend to develop diabetes at different levels of body fatness. For example, people of South Asian descent do not need to gain as much fat to develop diabetes as people of European and West African descent. Together, this suggests that each person has a “personal fat threshold“, and if they gain fat beyond that threshold, their fat tissue is no longer able to contain fat effectively, it spills over onto other tissues, leading to insulin resistance and a high risk of diabetes.
41. Mendelian randomization studies on the impact of insulin level on body fatness
Mendelian randomization is a genetic technique that can help to understand the relationship between risk factors and health conditions. Several such studies have examined the relationship between insulin secretion and body fatness. The first two reported no effect of insulin secretion or fasting insulin level on body fatness. The third study reported that insulin secretion increases body fatness, but that this mechanism only accounts for 1-10 percent of between-person differences in body fatness.
Thus, all Mendelian randomization studies to date suggest that the impact of insulin on body fatness is either small or nonexistent.
42. Of the three published studies funded by Taubes’s organization the Nutrition Science Initiative (NuSI), at least two, and possibly all three, refuted the carbohydrate-insulin hypothesis
The first study was a tightly controlled metabolic ward study reporting that metabolic rate slightly and transiently increased, but fat loss actually slowed, on a very-low-carbohydrate ketogenic diet (2% sugar), compared with a high-sugar (25%) higher-carbohydrate diet of equal calories. This study also reported that the very-low-carbohydrate diet did not increase total energy levels in the bloodstream, as claimed by Taubes’s model.
The second study was a one-year randomized controlled diet trial (DIETFITS) reporting that a low-fat vs. low-carbohydrate diet led to the same degree of weight loss and the same metabolic rate after one year, and baseline insulin secretion did not predict who did better on which diet.
The third study reported that a very-low-carbohydrate diet led to higher calorie expenditure than a low-fat diet in people maintaining weight loss. However, a reanalysis of the raw data suggests that the effect may be an artifact of faulty data.
Exercise
43. Exercise tends to cause weight and fat loss in people with excess body fat
This has been demonstrated by dozens of randomized controlled trials, although the findings are not always consistent across individuals or trials, probably because many trials use modest exercise interventions and people adhere to them poorly. Meta-analyses (studies of studies) tend to report that exercise causes weight and fat loss, including the most recent meta-analysis of 36 studies.
Greater volume of exercise, particularly when supervised to ensure compliance, causes greater weight and fat loss. In one randomized controlled trial that assigned overweight volunteers to different exercise amounts, greater amounts led to more body fat loss, with the greatest amount leading to a loss of 12 lbs of body fat over eight months relative to a group that did not exercise (175 min a week on a cycle trainer, treadmill, or elliptical).
Thanks to Adam Tzur of Sci-Fit for references.
44. Exercise increases insulin sensitivity
This has been demonstrated by many randomized controlled trials. Here are a few review papers of the evidence.
45. Sedentary behavior causes insulin resistance
Athletes have high insulin sensitivity, yet ten days of inactivity markedly reduces it. Five days of bed rest markedly increases insulin resistance in non-athletes as well. Reducing daily walking time also promotes insulin resistance in only two weeks.
Food reward
46. Fat and carbohydrate both cause dopamine release and increase eating drive, particularly when combined
The gut senses fat, carbohydrate, and protein, and this causes dopamine release in the brain that stimulates motivation and learning. This has been extensively documented in animals, and in humans it has been replicated for fat and carbohydrate.
The combination of carbohydrate and fat causes greater activity in dopamine-responsive brain regions, and greater eating drive, than either alone. To visualize this yourself, imagine how much you would want to eat regular ice cream, vs. fat-free ice cream, vs. unsweetened ice-cream.
47. The most commonly/intensely craved foods combine carbohydrate and fat together, while foods that are high in sugar or fat alone are less commonly craved
This should be common sense (think of ice cream without the sugar or fat), but it has been extensively supported by research. Foods rich in fat and carbohydrate together stimulate eating drive more than either alone, measured both by brain activity and people’s willingness to pay for the foods.
Studies on cravings and food addiction have found that foods combining carbohydrate and fat are the most likely to trigger cravings and addiction-like behaviors, including savory foods that contain no sugar. The most commonly craved food is chocolate. Foods that are only high in sugar or fat can also trigger cravings, but it’s less common.
48. We eat more food when it tastes better
This is common sense, but here are some references to scientific studies just in case anyone doubts it.
Rigor
49. Calling for more rigor in science does not necessarily make a person rigorous
Science has flaws at many levels that make it less efficient and effective than it could be. However, using the concept of rigor selectively to criticize findings you don’t like is not rigorous.
Those who genuinely care about scientific rigor should be promoting the most effective tools we have for increasing it: Study preregistration, open data, proper application of statistics, conflict of interest regulations and disclosures, replication, meta-analysis, and Registered Reports. They should also be promoting the organizations that develop and advance these tools like the Center for Open Science. The most effective tools for increasing the rigor of science are coming from within science itself– designed by people who understand how science works. Non-scientists can help by learning about these efforts, promoting them, and pushing for their adoption.
50. Expert reviews of Taubes’s writing have uncovered extensive misuse of evidence
Gary Taubes has an extensive history of misrepresenting source material to benefit his narrative. The most detailed fact checks of his books were written by Seth Yoder at The Science of Nutrition, and the results are frankly disturbing. My own review of The Case Against Sugar also uncovered evidence misuse and a general unwillingness to cite relevant scientific evidence and apply logic appropriately.
51. The Nutrition Science Initiative (NuSI), with Gary Taubes at its helm, attempted to interfere with the research that it funded once unfavorable results came in
Initially, NuSI assured researchers that it would have “no control over research design, conduct, or reporting”, which was essential due to Taubes’s conflicts of interest. NuSI asked lead researcher Kevin Hall to communicate this to me, which he did in 2012 and I documented on my website. Once the first results came in undermining the carbohydrate-insulin hypothesis, NuSI broke its agreement and began interfering with the scientific process, eventually causing the resignation of lead researcher Kevin Hall. Hall concluded that “Potentially conflicted funders should not be involved in the conduct, analysis, or reporting of research. NuSI’s meddling in the EBC study violated both the final contract terms and our verbal agreement at the outset of the study.”
Miscellaneous
52. The most fattening diet in animals and humans is human junk food, and its fattening effect cannot be explained by carbohydrate or fat alone
In rodents, the most fattening diet ever tested is the “cafeteria diet”, which consists of a buffet of calorie-dense, tasty human foods like condensed milk, chocolate chip cookies, salami, cheese, banana, marshmallows, milk chocolate, and peanut butter. When rodents are given a choice between the cafeteria diet and their normal low-fat rodent pellets, not surprisingly they gorge on the human foods and virtually ignore the boring pellets. Other species quickly grow fat on calorie-dense human food as well, including raccoons and monkeys. Humans with ready access to a variety of calorie-dense, tasty foods also overeat and gain weight rapidly.
In rodents, diets that are only high in fat or only high in refined carbohydrate (including sugar) are not nearly as fattening as the cafeteria diet, demonstrating that neither fat nor refined carbohydrate alone can explain its full fattening effect. Similar findings are observed in humans, where increasing sugar intake alone isn’t nearly as fattening as the cafeteria diet.
53. Obesity has a strong genetic component
This has been demonstrated by many different studies in humans. A meta-analysis (study of studies) of 115 such studies concluded that 75 percent of the difference in body fatness (BMI) between people is due to genetic differences. Note: this is perfectly compatible with the idea that diet and lifestyle changes are responsible for the high prevalence of obesity in the 21st century; these studies describe the genetic component of differences in body fatness between people in a given environment. If we all live in a fattening environment, those of us who are genetically susceptible to obesity will develop it. Without a fattening environment, these genetic differences matter less and few people develop obesity.
54. Synthesis of fat from carbohydrate is negligible in humans eating typical Western diets
The scientific term for this is de novo lipogenesis, and although the pathway exists in humans it only converts a tiny fraction of ingested carbohydrate into fat under realistic circumstances (typically less than 1 gram per day). In contrast, most people consume 50-200 grams of fat from the diet. See this review paper for the evidence, or the physiology textbook Metabolic Regulation, pages 200-201.
55. Partially suppressing the release of fat from fat cells (lipolysis) does not cause fat gain, hunger, or a slower metabolic rate
A drug called acipimox inhibits lipolysis, or the release of fatty acids from fat cells, mimicking the effect of insulin. As a consequence, free fatty acid levels in circulation decline (this is a key circulating fuel in the bloodstream). The carbohydrate-insulin hypothesis predicts that this should lead to fat gain, hunger, and a reduced metabolic rate. A 6-month placebo-controlled randomized controlled trial of acipimox showed that the drug reduces circulating free fatty acid levels by 38 percent but has no impact on body composition, calorie intake, or metabolic rate.
56. The fat tissue of people with obesity releases fat at a greater rate than that of lean people, not a slower rate
The most informative study to date on this found that the greater a person’s fat mass, the higher the total rate of fat (free fatty acid) release. Thus, nothing is “trapping” fat inside the fat cells of people with obesity– to the contrary, they release fat from their fat tissue at a greater rate than lean people.


”The weight loss version of the rice diet was low in calories, supplied over 200 grams of mostly refined carbohydrate per day, and patients also increased physical activity. ”
That`s an interesting study. In this study the authors wrote that the initial diet was a low calorie diet and that in the beginning 90-95% of the calorie intake were carbohydrates (rice, fruits). But 1 month after they added some vegetables and meat after that. Still 200 g carbs here after this modification? How long did they consume 90% of carbs or over 200 g of carbs? Thank you
Just wanted to say “wow”. This is an amazing list. Thanks for creating this list!
Ayland Letsinger
Doctoral Candidate
Texad A&M University
You are head and shoulders above all other science guests on JRE for doing this. Thank you for sticking it to Taubes.
Brilliant work Stephan. Well done on presenting the current state of the evidence.
This is great. Maybe you can find him a reference on how to pronounce your name?
I believe you should be looking in the DNA, allees have a different number of recombinant DNA. This could be a determination of how our bodies store fat. If your ancestry has suffered prolonged starvation then your going to increase you body fat fast as your body could have adapted to suit your surroundings.
Before I watched the interview, I did not side with either person. If Dr. Guyenet had been more willing to explain the mechanisms or reasons as to why the studies were definitive to him (after all, correlation is not causation and many studies are deemed invalid at some point), I would have been riveted. But just reviewing the abstracts doesn’t help me understand why the study should be reliable. Gary does have some good questions and I think the good doctor needed to step in and teach! Personally, I am hesitant to believe that we have discovered 10% of the genes (I studied genetic engineering briefly). It is foolish to be so confident – look back at history… remember when we thought exons were useless? An expert is always a student, always questioning with a critical eye and always teaching.
It is interesting that you use “correlation is not causation” as an argument here, given that the majority of Taubes’ argument is based on the very same type of observational data that we typically criticize in that fashion, whereas the majority of Stephan’s arguments were based on randomized controlled trials, i.e., specifically the types of studies where we CAN determine causation. It is not at all clear to me why Taubes gets a pass here.
This is just plain awesome. It’s like the entire summary of everything you need to know about fat loss in one article.
I’m bookmarking this to read twice a year at least to course-correct when my brain starts fooling itself about what I’m eating and its effect on my body fatness.
OK, you had more studies than Taubes and your certainty helped your case but it all seemed to miss the point when looking at epidemics in obesity and T2 diabetes.(already you have a criticism of Sarah Halberg – her results are tainted…) Those things are actually happening. Why?
Given that there will be, or there are, studies to refute your conclusions it seems prudent to say, no matter your opinion of Taubes, that eating high calorie high carb diets make you fat. Eating high protein/fat diets can also make you fat but will reduce overeating and make normal weight easier to maintain. There are mountains of evidence to prove this and the anecdotal evidence (right, not scientific) is overwhelming. So many people are so much better eating low carb it can’t be ignored.
At the same time the large agricultural companies and Drug companies, along with the vegetarian/vegan follows have a financial and political angle which can’t be ignored. Vegans have turned a political/moral ideology into a diet.
We have the experience of having accepted the studies and ruined the health of the nation. Scaremongering cholesterol and heart disease, eliminating healthy fats from diets were wrong and millions paid a bad price.
That has been my take also. I think there is a lot of value here in the critiques of Taubes and yet I have this nagging worry that for all the truth that correlation isn’t causation, there has been a shift in public health pressure in recent decades against animal fat, red meat, etc, and yet the T2 diabetes issue keeps getting worse. Sure, some sugar consumption has fallen but from still relatively high levels. And the current veganism drive could cause fresh problems if the calorie intake issue is not borne in mind.
A point that I think Taubes is at fault on is paying relatively little regard to what Stephan says about exercise in burning calories. I find it downright bizarre that exercise cannot play a part in regulating insulin resistance here and I would be keen to see more evidence around this.
Second, exercise and a training regime can possibly help a person to develop greater willpower, and this plays right to Stephan’s point about how our brains can stimulate cravings that can cause problems. So maybe a takeaway from this is that exercise (and maybe other disciplines) can help steer us away from destructive dietary behaviours).
I felt, listing to this JRogan show, that Stephan came across as a bit arrogant at times but then when debunking an opponent, a certain amount of tension is bound to surface. I am certainly going to follow this debate further and read Stephan’s book and blog in future.
Brgds.
The Taubes/Atkins diet’s drawing power is its simplicity. Exercise and calorie counting are derided because carb elimination is ALL that is required for HFLC plans to work.
This extremely simplistic plan fails once the dieter leaves it. There is no Plan B for eating carbs and exercising once the weight is lost. Most Atkins dieters relapse into their former habits and regain the weight. That’s not Taubes concern.
Great work. Truy fascinating to see it all in one place like this, in a condensed format. You did well in the podcast to present core ideas and not let Gary interupt you too much with Brothers Grimm tales. Sadly this podcast format is not the best to find truth, it gets rather personal. Scientists, doctors…were on your side already and the layperson will stick to his or her bias based on who was “the prick” or “condescending” in the debate according to them, but that’s fine.
Good work. Though I fail to see why you evidently serious nutrition people give agenda-driven journalist Taubes this importance. Ridiculous.
Till research stops fetishing on weight loss, it will never get clarity and reach the holy grail of congruent nutrition science. And when that is done we will have done to death the present toxic carbophobia monster.
My thoughts exactly. Where did Taubes get his “science” background? Doing a master’s in engineering?? One thing is to have a critical eye for spotting faulty science, another thing is to actually do science. NUSi tried and didn’t work out….
These trumpian personalities take excessive amounts of oxygen from the atmosphere we live in…
Guyenet: 10, taubes: 0
Great work Stephan
Excellent review of the current scientific evidence!
So, if you were to create a top ten list of rules people should follow to avoid cardiometabolic disease in our current highly processed food environment and dysfunctional eating and exercise habits, what would those ten rules be (distilling down the long list of 56 down to the most important 10 that people could actually focus on)?
I’ll guess:
1) Eat variety of nutrient dense, whole foods to satiety without paying too much attention to macronutrient composition except higher protein.
2) Resistance train once or twice a week and be physically active, preferably outside for 30+minutes a day.
3) Sleep regularly, 7-8 hours in a dark, silent, cool (~18 degrees Celsius) room, avoid stimulants (coffee, alcohol, chocolate, screens…) in the afternoon/evening.
Hi Stephan,
Just wanted to say that if you ever discuss with Gary again and he keeps mentioning how every study who refutes his hypothesis is because the researchers are “phase locked” into their position… Well, ask if the fact that he’s refuting all the studies that show his position is not correct, is that not a prime example of being “phase locked”.
It’s all he did on the podcast. You pointed to studie(S) and he said they were all bad because they were biased. That’s not an evidenced based argument. It’s an ideology!
FYI. I did not know who you were before this talk. I have read Gary’s book, found the info on eating well good. Although I always found his position on what causes people to get fat to just be an idea without actual evidence. Now I know why. It does not exist.
I have a question.
After listening to your arguments in JRE, what I got out of you was that you are basing the calorie intake on the influence of body fat.
In a part, you said that obese people consumed 15-30% more calories. So it doesn’t matter if they eat more carbohydrates or more fat.
So in study 52. you say that you need cafeteria diet to be obese and with only fats or only carbs you don’t get as fat. I would guess that if you want to state that this fact is true, the subjects were consuming the same amount of calories, so doesn’t that mean that the type of food you eat DOES make matter?
On high carb low fat/low carb high fat you will spontaneously eat less than on cafeteria diet. (Eating “Ad libitum”=as one pleases, which is what happened in referenced study).
Hi Zisis,
Reference #52 is about studies where the subjects were allowed to eat as much food as they wanted. They ate more (and thus gained more weight) when the food was more palatable.
Stephan isn’t claiming a hyperpalatable diet (such as the cafeteria diet) is *necessary* to become obese; just that it makes it a lot easier to consume enough calories to do the trick. If someone was able to force themselves to eat just as many calories’ worth of healthy food, they would become just as obese.
Hello Will and Ondřej,
Thanks for clearing this up. Sorry for the bad grammar but I didn’t know what I can’t edit the reply 🙁
I didn’t have the time or the knowledge to go through the article that was referenced so I assumed that they were consuming the same amount of calories.
So “cafeteria” food made them consume more food thus consuming more calories thus becoming more fat. OK so does support Stephans argument.
I learned a lot by listening to your talk on Joe Rogan’s podcast. I’m digging into your site and enjoy your work and research. Nice job sir!
Your sugar intake graph (#17). Do you think hidden sugars are included? Sugary cereals, sugar in ketchup, sugar in sauces, sugar in pickles, sugar in bread, maltodextrin and all the aliases, etc. Or is it sugar as a stand alone product (store bought sugar, honey, karo syrup, etc.). People don’t cook at home anymore, and how the heck would someone know the sugar intake of people who eat out all the time, or eat processed foods? Did someone follow people around? These data seem dubious at best.
Yes, that is all included. All sucrose, high-fructose corn syrup, honey, and other sweeteners.
As I understand it, the USDA data are production-based. It’s not from following people around but from annual volumes of foods going to market, from which the macronutrient profiles are calculated. It undoubtedly overstates actual consumption, but it is useful for historic trends, especially the macronutrient ratios.
Thanks Stephan! That’s a great list of references.
I just wanted to thank you for sharing your “low carb” phase. I used to feel so silly for falling for it also after reading Taubes’s book. The fact that you, and other people I admire deeply, also went through a period when they thought low-carb was THE diet, makes me feel better for having been so uncritical of GCBC.
I went over two years on a keto diet! Lost weight (gained fat/lost muscle), my cycling and running went to hell (so much for fat adaptation), my LDL went through the roof, all the while thinking it didn’t matter cause keto would make me live to 100 free from all diseases…
Thanks specially to you (your blog, your book, and your peers) I’m back on a whole foods healthy high carb low fat diet (I try to emulate that of the southamerican traditional cultures) and feeling, performing, and looking better than ever!
Keep up the good work and thanks again.
This is an outstanding source of studies! I found this site from your podcast with Joe and Gary. I learned a lot from the podcast. Gary was a great punching bag and a typical useless baby boomer that needs to hurry up and stop stealing oxygen.
Wow what an assine comment. Whether you agree with Taubes or not, wishing him dead is beyond reason.
Stephen, I don’t see any responses from you in the comment section, so maybe you don’t respond to comments, but I’m very interested in understanding this better, as health and nutrition are very important to me.
I agree with you that calories absolutely do matter, and I want to make sure I understand your position. Do you think calories are the only thing that matter? So if person A eats healthy food and person B eats unhealthy food, but they both consume and expend the same number of calories, no weight gain should occur, correct? You believe that the mechanism by which unhealthy food causes weight gain is purely through food reward, meaning it causes you to overeat? Is that right? Does that mean that if someone eats unhealthy food their entire life, but stays within the right number of calories, that no weight gain will ever occur for them? Will the unhealthy food cause any other problems for them? The studies have convinced me that in the short-term no difference in weight will occur. I think you can lose weight eating only Twinkies if you eat few enough calories, but I think the Twinkies may take a toll on your body in the long-term. What do you think? When I first read Good Calories, Bad Calories, I was convinced Taubes was right, but several years ago, some of the things you shared caused me to update my views—mainly the studies showing that people on low-fat diets lost weight similarly to those on low-carb diets. All of those studies are short-term studies though. Do you think foods like sugar or refined carbohydrates may contribute to insulin resistance in the long-term? I guess that is a weaker version of the insulin hypothesis, that excess carbs lead to insulin resistance which leads to obesity. Also could genetics play a role in how much you may be affected by sugar?
Abe,
Congrats for your willingness to revise your view when faced with new evidence. I was more lucky because I found the right people to begin with. 😉
Regarding your question, using isocaloric diets (diets with same calories), protein causes a little more caloric expenditure than carbs, and carbs cause a little more caloric expenditure than fats. But when you go into ketosis, you also spend a few more calories, so maybe the difference between carbs and fat is actually very close to zero.
In the end, all these differences are rather small so *approximately* isocaloric diet will deliver same outcome, especially if they’re also equal in protein intake.
Please note that spending more calories because you’re in ketosis or because you’re eating a lot of meat isn’t a good thing. Actually it is probably a very bad thing. You’re probably spending more calories because your body has to work harder to deal with the diet.
Regarding long term vs short term, that’s actually the interesting question. There are no studies on this because it’s impossible to control and to monitor food intake for long term. But it’s fair to guess that processed foods and animal foods will tend to cause health problems, and the health problems will *eventually* cause poor diet compliance. You’re also much less likely to maintain an active lifestyle if you’re always feeling sick and tired.
It’s not clear if raw high fat plant foods like nuts and seed are likely to cause significant health problems. We know fried oils and saturated fats cause health problems, but we don’t know about raw high fat plant foods. We don’t know but surely it’s a risky choice.
Slowly digested high carb foods are not likely to contribute to insulin resistance in the long term, especially in the context of a good diet (a good diet provides you all nutrients that your body needs to function optimally). In fact you can cure insulin resistance with this kind of diet, as it has been proven in several studies (they’re on nutritionfacts).
Quickly digested high carb foods do cause some health problems, especially in the liver, but they’re unlikely to cause massive problems. It also depends on the context. A fruit juice is much healthier than a coca cola even if they’re both rapidly digested high carb foods.
Sure, genetics can play a role. But most of our ancestors were living in temperate regions and eating mostly plant foods. They weren’t Eskimos. Population relying primarily on animal foods like Eskimos simply can’t reproduce enough to spread their genes.
I hope I’ve answered to the most important questions.
Abe, basically we’re plant eaters due to evolution. You can’t feed a large population with mammoths and bears. There aren’t enough of these. On the other hand, you can feed a large population with potatoes and grains. And this is what actually happened over the recent history (the recent history is the only history that we really know). I recommend you look at the effect of the potato in north europe. Basically the entire post-1500 boom in north europe is due to the introduciton of the humble potato. It’s an high glycemic index plant food. Who cares if it’s absorbed too rapidly? It works. It has caused a massive growth in population!
Hi Stefan,
While I agree with your position on Rogan’s podcast, I found your attitude incredibly dismissive and absolutely condescending towards Gary. While your arguments may be right, I hope you learn to be more respectful and less arrogant in your future debates against more challenging opponents..
I suspect that you will abscond and shrivel at any criticisms towards your character – you seem to self-promote quite well on your website. In any case, if you do not decide to leave this comment on your website (as I suspect), then I hope at the very least you visit your nearest baker and eat a slice of humble pie.
Hi Jesse,
It’s been interesting to see peoples’ reactions to my delivery. You are correct that I don’t take him seriously; he has been peddling this hot air for over a decade and I just find it amusing at this point. You saw how poorly he was able to defend his position; if you were an expert arguing with someone like that, would you take him seriously? I also had to be very assertive with him to prevent him from talking over me constantly, which is what he does in every debate.
But the main point I want to get across is that nobody is more condescending or arrogant than Taubes. He refuses to call obesity and nutrition researchers “scientists” because he thinks we are so useless. He has called the entire field of obesity researchers “idiots”. He paints entire fields of research as blinkered automatons (“phase locked”) who can’t see beyond our noses, constantly making stupid “assumptions”, even when the evidence demonstrates he’s wrong. And he broadcasts these arrogant views to the public via his writing, stirring up hate toward the scientific community. I literally could have wiped my butt with a page from his book on camera and I still wouldn’t be as arrogant or condescending as he is. Is the scientific community supposed to sit there doe-eyed while he takes giant misleading dumps on us, or are we allowed to hit back?
I also agreed with your position Stephan but you don’t seem very self-aware and empathetic of the perspective of your situation from the outside view. To the majority of the audience on the podcast have no idea who you or Gary are, or the context of your relationship history nor of his track record. So someone in this position watching the podcast would have no reasonable assumption to believe Gary is the character you describe – as usually people with such characteristics generally manifest them more in line with, well frankly, the demeanour you came across with in the interview ironically enough. I’m talking about being rude and condescending. While I understand that from your personal perspective and knowledge of this person the disdain might be justified, you lost the PR battle as he came across as a being far more respectful in tone (Even if not so much in content as you state clearly).
You had this conversation in public, you made a huge mistake assuming your audience would also dislike this person and therefore think your tone justified. You should have walked your audience, hand holding, to the conclusion that this person was misrepresenting and dangerous to earn the right to be dismissive and rude in demeanour in their eyes in order to not turn people off your argument. You basically gave people a massive stick to beat your own back with, emotional dislike of your character, while trying to (presumably since this was in public) persuade them to your point of view.
I will say you did well to stay on track though, Gary really was quite annoying, regardless of the validity of his point, by making continuous appeals to long anecdotes. Ugh so hard to listen to.
Good luck in the future, and maybe try some self control for the first 30-60 minutes of the interview before you go for the jugular next time, facts aren’t often wholly convincing to audiences unfortunately.
Hi Bob,
I appreciate the feedback. I actually was surprised by the perception that I was being arrogant; I didn’t feel like I was being arrogant. How I felt was amused and assertive. As I mentioned I had to constantly parry his attempts to interrupt and talk over me, and I can see how that might have come across as disrespectful. I sat and listened patiently while he spoke at length, and interrupted him much less frequently than he interrupted me.
There was plenty of evidence of his arrogance in the debate, so I don’t get why he came across as more sympathetic. Maybe because of his stories or self-deprecating dad-humor? Maybe because I was punching down? He was taking dumps on the scientific community the entire time.
In any case, it is a learning experience for me. If I had known how people would react I probably would have tried to modify my delivery. There was no alternative but to be assertive with him though, people who have debated him previously have told me that he will just talk over you the entire time unless you shut him down.
My unsolicited 2 cents: I listened to the entire thing. On the science, it seemed to me (a definite non-expert, so, judge accordingly…), you came with an attack helicopter with rocket launchers and gatling guns, refuting Gary at every turn. Gary had, at best, an M16 (maybe a story telling M16). Seems like Gary hung his hat on 1 outlier study, the Pima, and Trinidadians, while admitting there are alternate explanations for the two populations. Actually, he had a cap gun, I don’t think he was able to seriously push back on anything you said.
I do think your snickers while Gary was talking was not a good “look”, so to speak. But I don’t think it was as bad as other people have said. Being assertive is not a bad thing. It is known, though, that Gary is somewhat a friend of Rogan (he’s been on at least twice I think, and Joe is a keto proponent, so sort of primed to believe Taubes) so you were, in some respects, going into enemy territory, at least among Rogan fan boys, so that might explain some of the more extreme reactions. However, Joe did seem fair to you.
It really was a shame about the combative tone, I bet many people didn’t make it all the way through. I would love to see just you on JRE. There’s so much more fascinating stuff in your book, that I think you could easily do the normal 2-3 hour Rogan podcast about the body’s energy homeostasis system and its dis-regulation. It would have been so much better to first have an episode with you, and then a discussion with Gary.
I didn’t think you came across as personally arrogant. Instead, it was arrogance on behalf of the scientific community. It’s not an effective debating tactic today, when science is in disrepute. Today, in a public forum, you have to be able to defend science and explain it in everyday terms. Taubes, with his background of science writing for the public, is a master at this — enough to abuse his powers. You, in some respects, have yet to try. For instance this “palatable” stuff: how does that translate into language that Rogan’s viewers (or even I) will understand? It seems the basic idea is that people eat more when the food tastes better. If you were to put it that way, you’d find the audience naturally agreeing with you; and even Taubes would find it hard to deny. If he tried, you could tease him: “what, you’ve never taken an extra bite of something that was particularly tasty?” But what then? Should a whole lot of us start eating tasteless gruel in order to get back to healthy weights? What about your old post extolling the savory aroma of bone marrow? Is that too to be given up in search of tastlessness? Are the overweight to behave like the old Puritans of Boston, who neglected lobster and oysters in favor of (as an old rhyme put it) “pease porridge hot, pease porridge cold, pease porridge in the pot nine days old”? Are there no innocent pleasures in food?
You two were the only ones with the full context about your relationships. For all we the audience know, he is fully justified in insulting the nutrition researchers of the past century. We also have the reasonable assumption that he’s not referring to all nutrition researchers. The thing you didn’t display is that you were there to educate the audience rather than take down Gary. I got the idea that taking down Gary was your primary reason for being there. On that front, I would have liked to have heard two things from you:
1. What is your thesis, precisely? In my mind, when I hear both of your hypotheses, I ask myself why both cannot be true? I’ve had to buy your book and listen a few times to figure out your objection, and it seems that you’re right that obesity is caused by sugar *and* fat being present at the same time. This is something I entertained on my own for a while after I learned that de novo lipogenesis is a very slow process. But I still don’t know exactly what you mean when you say that your brain makes you fat. The brain drives behavior, and your book reflects this in the advice you give. Are you saying that the brain signals your body to store fat or that it will drive behavior to eat to a particular point?
2. Engage on the idea that hormones are part of the regulation of fat stored on the body. It seems like you’re ignoring looking at the problem from this angle out of disdain of Taubes and not out of a rejection of it as a valid study. This is a valuable area for you to talk about or refer to people who can.
3. Find where the two of you agree and build on how you diverge from Taubes. The entire time it seemed that on certain points you two were talking past each other.
And there’s just considering that you’re taking your audience along with you to this understanding you want them to have. For example, the first time Gary interrupted you, I was right along with him. I didn’t get where you were going with this idea. It felt like I had joined the conversation in the middle of it even though I listened to the podcast from the beginning.
At the end of the day, you got a few bucks out of me for your book, so that’s a plus for you. However, Gary is still asking some good questions, so I can’t discount him entirely. I also don’t have a great impression of you because you haven’t actually come to terms that you conducted yourself poorly in the context of the situation. I hope you take this and try to become more patient for your audience. If you go back on Joe’s podcast solo, I will be happy to watch because I am curious about your ideas.
Joe’s audience definitely know who Gary is without a doubt. It’s pretty obvious that Joe has always had a low-carb bias. I think the backlash against Stephan is largely from those who agree with Gary’s low-carb stance.
Hi Stephan,
Thanks for your reply. I guess I came at the situation without enough context to judge the debate. From the brief exchange on Rogan’s podcast, it would seem that Gary’s arguments were absurd indeed.
I really respect you for publishing my comment. My criticism to your character was perhaps overly harsh and exaggerated, but you’ve proven me wrong and I respect you a lot for your response. I look forward to hearing more about your work.
This is exactly how I felt too. I celebrated as you defended evidence based obesity research relentlessly without softening your tone a bit. Gary’s track record on mocking obesity researchers and nutrition experts outrageously is looooong-lasting.
This time, as there was broader audience than just his usual carnivore/LCHF fans, Gary’s strategy seemed to be to play a role of a nice, common-sense guy and drop meaningless anecdotes; clearly he had no chance of winning on facts and scientific evidence. All he could rescue was empathy points.
As expected, his fans felt pity for him and therefore now attack you (ad hominem), not the facts or the points you made.
Stephan,
Excellent job, on many levels (content, clarity, assertiveness, and not losing your shit with having to listen to over 2 hours of gibberish), with your appearance on JRE and debating Gary “story teller” Taubes. Furthermore, this post does a great job of laying out the evidence. Keep up the great work
Great work Stephan. Taubes is ridiculous in every way. I too will be writing blogs on these subjects you mention. Anecdotally, I have intestinal failure, don’t eat anything, and only live off orange juice, Fortisip and Ensure prescription drinks, and although it comes with complications, I’m physically very fit, have low body fat, no diabetes, no obesity, no drugs, no caffeine, no insulin resistance, almost no water (due to my liquid intake).
Hello Stephan,
I have a very specific question that is a followup of what was discussed at the end of the podcast regarding insulin and carbohydrates.
I want to eat 3 big 450cals donuts. Does insulin handle them the same way if I eat them on an empty stomach and if I eat them after a full 1500cal low carb meal? Let’s assume that I am going to sit and watch a movie and not do any physical activity in both cases.
In other words, does insulin push carbs into fat storage faster if I eat them on an empty stomach and slower if insulin is handling the lower concentration of carbs because they are distributed with the other nutrients of the meal?
Hi Stephan,
Thanks for taking the time to debate on JRE.
I feel there is so much more we could have learned, if you were there alone and explaining your position.
Hopefully you can get back and do another extended podcast with just you and Joe.
Let me first say that I am a huge fan. I think you have basically taught me everything I know on the subject of obesity and nutrition. I also believe Taubes is a liar and a fraud who has done more bad than good. I was really rooting for you on the Rogan podcast.
Unfortunately, I couldn’t help but feel that much of what you had to say was reactionary to Taubes. There is a phenomenon in hockey where players “chirp” constantly at each other in order to get in their heads to mess with their focus so they have a bad game. I feel like you were on the defensive during the podcast and that Taubes was indeed in your head.
I’ve listened to you speak many times on various podcasts where you speak eloquently. As a lay person with no background in the field, you have a way of helping me understand complex concepts easily. On Rogan, I would have liked to see you focus more on explaining these concepts to the thousands of people listening that really know very little about nutrition. Let’s face it, very few people are actually going to look up those references. But the way I have seen you break down concepts before has been compelling in itself. By the way, you did do some of this, for example when you explained about human energetics, just not as much as you normally do. I think that if the conversation had been directed more at helping Rogan understand the concepts, rather than disproving Taubes, it would have made for a better conversation. I got the impression that Rogan was really open to hearing from you, but the conversation diverted in attempting to disprove Taubes.
I think mentioning that you yourself once followed a low carb diet after reading Taubes’ book was a good attempt to connect with the audience. I think “debating” Taubes could have worked if the disdain for him could have been left out and if you had looked at him as being your past self (as many still are) who believes that sugar is bad. I’m quite sure that if you met someone today who had just read Taubes’ book and believed every word, you would probably be patient and kind in explaining why it is not true.
I hope you keep perfecting your art Stephan because you have so much wisdom that the world needs to hear.
Sincerely,
Kelly.
I agree Stephan’s solo podcast would be much better for JRE audience, and for explaining the concepts and touching upon sleep and activity as well. Regarding the duel, there was a recent JRE duel between Chris Kresser and Joel Kahn MD. So again, a layperson vs. a scientist. Joel Kahn was super professional, respectful, didn’t promote his specific diet too much and did everything people suggest Stephan should have done, but there were still many insults in the comments towards him.
Gary always provides his credentials, and has spent a great amount of time in his research. Both are well versed, regardless of who you prefer, the message is….”eat better to prevent chronic illness”.
Let’s not lose site of what they both represent, and that is being more aware of what we eat. Good debate, at the end of the day, they both are on OUR side.
Hi Stephan,
I am halfway through the podcast and wanted to review your references and provide feedback in a more constructive manner than you’ll likely find in the YouTube comments.
As a longtime JRE listener, I have been awaiting podcasts which feature guests to counter some of the contrarians so as to provide counter-narratives. I think it’s healthy to not only seek out evidence from both sides, but to digest that evidence from both parties in tandem – so that claims can be countered on the fly. Reading long-form op-eds and such on both sides of an issue don’t allow for the interspersed rebuttals necessary to confront assumptions before they lay foundations for misconception.
That said, I read your response above and saw that you felt your tone, demeanor and behavior on the podcast was assertiveness mistaken for smug condescension because the audience was unfamiliar with your history and how you feel he has maimed your field’s pursuit of truth by myopically pursuing evidence which supported his existing beliefs.
I want you to know that as an individual who entered the podcast with an understanding of your history, the context in no way made your delivery seem justifiable. You were in fact smug, condescending and, frankly, unbearable. If you had been prosecuting the perpetrators of global-scale child sex ring, you would have still come across as unbearable. That is not to say that you are, in fact, unbearable. It’s your delivery, which can easily be amended, but I fear that if you don’t take this criticism to heart you will not only fail to change minds, but will turn people away from your research and way of thinking because they just plain won’t like you.
You clearly entered the debate more prepared, and had a fantastic opportunity to demonstrate the quality of your arguments and what you perceive to be the frailty of Gary’s. That’s why I genuinely hope that you will not insulate yourselves with the comments from your supporters indicating that all you did was serve him his just desserts, and your “assertiveness” was overdue. If you truly self-reflect, I think you’ll realize that the listeners were not turned off by “assertiveness”. I hope you will internalize this and change course. You owe it to yourself not to turn people away from you and your research.
You’re doing great work. Please adjust your delivery so that people will actually want to hear about it.
Cheers,
Duffy
Hi Duffy,
Thanks for your feedback. I recognize that my delivery could have been better for reaching a wider audience, and that is disappointing. It is certainly a learning experience for me.
It’s rare to see someone respond to this sort of feedback and acknowledge an improvement area. Consider me a fan Stephan. Keep up the great work.
Hey Stephan,
I wrote you a long time ago about having a problem losing weight post-menopause. Well, last year I succeeded in losing 20 lbs doing a combination of the following:
1. IF – 16/8 and 18/6 model 6 days per week.
2. Calorie Counting of 1550 cals per day 6 days per week during eating window.
3. Every Saturday I eat whatever and whenever I want. This step has enabled me to stick to my eating plan.
This year I’ve only lost 2 lbs so far but I had a knee replacement on January 2 that has been challenging since I discovered there is no narcotic pain meds on earth that I can take. I did eventually end up on gabapentin for nerve pain after an ER trip because I hardly slept in January because of the pain. Doing well now. I’m 58 now and hope to lose another 15 – 20 by the end of this year, which may or may not be a good weight for me. I will re-evaluate when the time comes! I am back to lifting weights and doing low impact exercise.
I believe the cheat day (food reward) was key for me because it still causes me to eat less 86% of the time.
What are your thoughts?
Rita
I lost 50 lbs in 6 months at about your age. This fixed my diabetes. But in losing the fat I lost a lot of upper body muscle mass too. It’s taken 10 years and 10 lbs weight regain to get it back. The older you get the harder it is to build muscle.
I in no way think Gary has it all right, and I know he can be difficult.
But your actions were disappointing. Like I feel about all all my past regrets, I hope you have a chance to make up for this “talk down to” moment.
I imagine it must be frustrating to have a journalist pretending to have authority on a subject that he doesn’t understand, especially since he’s calling the qualified researchers corrupt—an accusation made without evidence.
I know talking down to people is rude, but to be fair, he’s talking down to a charlatan who’s misleading millions of people while ruining the reputation of perfectly well-intentioned nutrition researchers. I’m not sure what the proper way to treat that kind of person is when debating him like that.
Congrats for your performance but why did you put yourself through this? Quite painful to listed to this old, delusional, uneducated, illogical and biased individual. Like trying to teach a chimp math while all he wants to do it to steal your banana. These type of people will exist and there is nothing nobody can do to change their way of thinking. Leave them avoid sugar and vaccines and pray to god … Keep up the good work and all the best!
I’m glad he did. I work in the fitness industry and there are a lot of laymen out there who intuitively side with Taubes. I think it’s important to bring on a qualified expert to debate those claims and explain how things actually work.
It’s not about teaching the chimp math, it’s about engaging with the chimp so that you can teach the onlookers math… and that way the onlookers won’t let chimps steal their bananas quite so easily in the future? That’s a garbled analogy. But anyway, I think it’s important that guys like Stephan step up to debate these influential but incorrect diet journalists.
I listened to the podcast in its entirety. A lot to digest (pun not intended, but funny)…
Putting the exact mechanism aside for a moment (brain vs. insulin hypothesis), can anyone explain how the following is *not* an endorsement of a lower carb approach to eating?:
1) Eating excessive/large quantities of carbs is a lot easier than eating excessive fat
2) Consuming high quantities of carbs corresponds with people feeling hungry more often
3) As Stephan himself admits, fasting is easier when eating low carb/higher fat
In other words, even if it is correct that intake of more calories is ultimately what causes weight & fat gain, it still seems obvious that by eating a higher percentage of fat (and therefore a lower percentage of carbs) in one’s diet, he/she would create conditions where it is more difficult to overeat! Isn’t this really the point?
The fact that fat allows for us to become satiated faster seems to me the most damning evidence against eating high carb. Couple this with the paleo diet argument that we clearly have increased our carb intake as a species only relatively recently (post agricultural revolution) and would therefore not have been able to evolve that quickly into an entirely new way of eating, and it almost seems absurd to me that there would be a serious argument against lowering our carb intake.
Seriously, what am I missing here? Could Stephan or anyone else explain this to me? Thank you in advance for any explanation you can provide.
Greg
Point 1) is probably the opposite. After all, it’s the fats that increased the most in SAD or any Western diet. Figure 18 above.
2) and 3) It’s probably easier to have low frequency meals on LCHF. But meal timing is basically unimportant compared to calories and if you lat people eat what they want, both LCHF and HCLF achieve similar fat loss.
Yeah what am i missing too? When people eat more carbs, sugar, grains etc they get sick. You remove those from your diet and ailments go away along with inflammation. Why would someone dispute this? Why would you say sugar is ok? It’s maddening
Hi Bob and Greg,
I just want to reiterate that I’m not saying refined sugar is OK. In fact, I emphasized several times during the debate that I think it (and other refined carbohydrate) contributes to obesity, and probably diabetes and cardiovascular disease. What I was trying to demonstrate in the debate, and in the refs above, is that sugar is not a steamroller that outweighs everything else in the diet and lifestyle. That is the argument Gary made in his book The Case Against Sugar.
I agree with the points made by Ondřej above. As I emphasized in the debate (literally the second point I made), low-carbohydrate diets are a valid tool for controlling appetite, body composition, and blood glucose. But that doesn’t make them the only valid tool in the world. The hypothesis that carbohydrate is fattening and fats are slimming cannot explain the fact that similar effects on appetite and body fatness are observed on both ends of the macronutrient spectrum. I’m trying to provide a better model that can explain all the evidence. But my model doesn’t imply that low-carbohydrate diets are ineffective or that we should start eating refined sugar.
Stephen,
Thank you for your time in addressing my question. I admire your involvement in this discussion thread, and your willingness to promote an exchange of questions, ideas, and information.
Some quick context for where I’m coming from… I don’t claim to have a detailed grasp of the cellular-level mechanisms of fat storage and metabolism. However, as a long-time triathlete and runner, I have had much experience with maintaining/managing weight and body composition. If I were to distill my experiential learning over the last three decades or so, it would be as follows:
1) Eating low carb while in periods of little to no training is, for me, the ONLY way to prevent significant fat gain.
2) During periods of heavy training (over 50 mi/wk of running and/or over 150 miles/wk of cycling), I can gain fat if I eat carbs freely. Moderating carb intake promotes fat burning as fuel for training, and I notice the more even availability of energy throughout the day.
3) Starting my day with a very modest breakfast high in protein and fat (e.g., bacon, eggs, avacado, nuts, along with veggies or berries naturally low in sugar content) guarantees that I feel very little or no hunger throughout the morning and early afternoon, with plenty of energy.
4) Restriction of bread, pasta, cereal, pizza and other carbs makes my digestive system MUCH happier.
I realize this is just anecdotal, and I also know you are trying to look beyond “telling stories” toward hard evidence. But I can tell you this is impossible evidence for me to ignore, personally. And I am absolutely sure there are many more out there who have experienced the same effects I describe above. All of this leads me to a simple question:
Isn’t it possible that a) calories matter, and any combination of macronutrients will ultimately lead to the same weight gain or loss, depending on the quantity of calories consumed, AND b) it is easier to maintain a lower level of calorie consumption when eating less carbs and more fat, due to the beneficial effects of such a diet on insulin and blood sugar? This would seem to me to be the basis of a “compromise” between Gary’s position and yours.
I’d appreciate your thoughts on this. Thanks!
Greg
There are many ways to skin the cat Greg. I trade counted exercise for counted food and maintain weight on any macro. Both carbs and fat support the exercise. It’s ALL fuel for the fire.
Sedentarism carries its own risks. Do more and eat more. Be like a Paleo and MOVE.
Thomas,
I am in complete agreement about movement being essential to health. And yes, all macros are fuel for the fire. But I would say that when I am running over 50 miles a week that is a fair amount of movement. And yet, it is not enough to burn off everything if I just eat whatever I want! As I stated, it is quite easy for me to *still* gain fat if I don’t ALSO restrict/monitor what I eat. And that is where the macro ratios are important, as higher carb intake promotes more eating!
I think one of the biggest problems with this whole low carb “controversy” is that there are always going to be people like you who are fortunate enough (and young enough?) to be able to easily burn off whatever you eat. People have different resting metabolic rates. Therefore, some assume that exercise is a sure-fire way for anyone to maintain an ideal body weight and composition. With my 50th birthday a month away, I can tell you that just isn’t the case, even if I might have agreed with you 25 years ago.
Greg
I’m 66 Greg, and have been counting exercise and food calories for the last 12 years to maintain my weight. My BMR tracked pretty close to Harris Benedict when I was counting carefully. I’ve gotten sloppy and undercount my food now, but can maintain on a daily calorie deficit of about 100 in the way that I count.
My exercise burn numbers are 70 cal per mile walking and 35 per mile biking. To get near Cordain’s typical Paleo level of activity
(https://www.sciencedirect.com/science/article/pii/S0033062011000648)
I need to walk about 12 miles a day, or bike 24. It takes time but I’ve done it for 12 years.
My motivator is the diabetes that I had. Restricting the calories and carbs to lose 50 lbs cured it, but it is the walking and biking that has maintained my weight. I chose exercise so that I could eat anything I like and not have to live in what felt like perpetual starvation at 1500 cal/day.
Greg, a high carb diet is like Stephan’s Cuban rice/sugar example. There is very little fat present, and it’s not very appetizing. This is not the same as the cafeteria or junk food diet. Look at the nutrient composition of a bag of snack chips or cookies and you’ll find 50% fat/50% carbs, with virtually no protein. It’s the combination of the two macros that makes them appetizing, not just the carbs.
Thomas,
It is not necessary to point to the extreme Cuban example to illustrate a high-carb diet. In fact, it is not a good representation at all. There are countless foods in the current American diet that are much higher in carbs than fat, while being very appetizing and prone to overconsumption. Some examples:
*Breakfast cereals – a HUGE one, and one that spikes blood sugar in the morning, paving the way for being hungry the rest of the day
*Various snack foods – this includes corn and potato chips, pretzels, popcorn, etc. Note that even where these may have fat content, many have substantially more carb calories than fat, and are still consumed in high quantity.
*Pasta, potatoes, bread – again, with minimal addition of fat (usually via butter) these can be quite appetizing and are very easy to overconsume.
These are just the first few examples I could list. There are many more. The irony is that many foods in these categories are marketed as being “healthy” precisely because they contain little or no fat, so people feel free to eat as much as they want!
Greg
I became obese and diabetic overeating breakfast cereals Greg, so I’m no stranger to overeating carbs. I counted carbs for several months and eliminated the diabetes. I’d recommend John Yudkin’s book “This Slimming Business”to understand the mechanics of low carb dieting, written by a vetted nutritionist. Yudkin counted carbs, and his book contains several pages of his”carbohydrate units” for dietary planning. His method forces calorie reduction by cutting high glycemic carbs to a minimum. However, he does not push it to the point of ketosis, nor does he eliminate any foods. Starches and sugars are allowed, but are very restricted.
Hi Stephen,
I enjoyed the podcast a lot. Gary’s obnoxiousness definitely outweighed any smugness on your part.
I was wondering what you think of the hCG diet and if it has any implications for obesity research. I have a degree in molecular genetics and I’m quite curious about the mechanism of hCG but it seems to be unaccounted for in obesity research although it definitely works for weight loss.
Hopefully we will hear you on JRE again…. alone next time 🙂
The most intelligent defense of the Taubes/Atkins low carb position was done 70 years ago by Yudkin. His concept of carb unit counting is central to the ADA’s carb counting methodology. It works especially well for diabetic blood sugar regulation, though Yudkin’s original intent was weight loss dieting. Taubes/Atkins uses Yudkin’s concept without articulating or crediting it.
The endless arguments regarding which weight loss diet is best never address the issue of long term weight maintenance. There are many effective methods to lose weight quickly. This ca six month effect doesn’t need to devolve into a street fight over which one of them is best. All effective methods require rigorous compliance. Leave it at that.
Neither you nor Taubes address the problem of weight loss maintenance directly, though you certainly hit on the reasons why it is nearly impossible. IMO the rigor needed to lose weight has to be maintained to offset the higher weight setpoint, and the brain NEVER forgets what that setpoint was.
Thanks for all the material. I especially like the USDA trend graphs, which reveal different things every time I look at them. This time it is our steady shift towards the “cafeteria diet”. We eat what we like to eat, which easily leads to eating more than we need to.
After listing to your talk, I just checked two references, and both fail to support your arguments.
Cuba: The primary energy sources were not (refined) sugar and rice, but *sugar cane* and rice. We know that there is a difference between chewing sugar cane and eating or even drinking refined sugar. (Same applies to honey). Also, energy intake was reduced by 1000 cal (30%) and share of active adultes rose by more than 100%. So even if the sugar cane had been fatting, this got over-compensated by the *massive* changes in energy and activity. Especialle since carbs were already 65% prior to the crisis.
24. “In human randomized controlled trials, sugar is only fattening by virtue of its calories” is a long refuted Sievenpieper (i.e. Coca Cola) meta-study that itself draws on flawed sugar industry studies from the sixties.
The high-carb idea can obviously be excluded (many traditional high-carb societies). The high sugar idea remains valid. The very few traditional relatively high sugar societies use mostly fruit or perhaps honey, and they use it only seasonly, not constantly.
Hi Andrew,
Do you really think that people were chewing on whole sugar cane for 28% of their calories? I’m not sure that would even be possible. Have you ever eaten sugar cane? I’m pretty sure they were eating sugar, not gnawing on canes.
Regarding calorie intake and activity, you’re making the same point I made. They matter, and they override the effect of sugar on body fatness, contrary to Gary’s claims.
Regarding your second point, you can’t just point to a person’s conflict of interest and dismiss their evidence without finding substantive flaws in their study. Sievenpiper has received funding from a variety of sources, including several food industry sources, but you act like he’s in the soda industry’s pocket. I’ve interacted with him, read his studies, and I don’t think that’s the case. His meta-analysis doesn’t say that sugar isn’t fattening– it says that it’s fattening because of its calorie content. This isn’t exactly controversial, and it doesn’t exactly make sugar look good.
As for your other comment, I didn’t approve it because you were just slinging mud.
Hi Stephan,
Regarding Cuba: Fact is, the study you cite only mentions sugar cane. There is no information on refined sugar, sugary beverages etc. The study also doesn’t mention the baseline sugar consumption. It is quite possible, for instance, that soda consumption went down, because Cuba couldn’t import Pepsi/Cola anymore (the study mentions that food imports collapsed).
(Sugar cane is also consumed e.g. in the Philippines. Studies show it has very different dental health and metabolic impact than pure refined sugar).
Regarding calories/activity: My point is that Cuba is an extreme case / outlier that doesn’t support your argument. We’re talking about a permanent, multi-year 1000 cal reduction and an increase in activity share by 100%. This obviously doesn’t refute the fructose/sugar hypothesis at +/- constant energy intake.
Regarding Sievenpiper: I mentioned several examples in my second post, which you simply deleted (instead of editing it). I read not only his meta-studies (which have serious selection flaws, favoring industry-funded studies), but also every single individual study to really understand what he was doing.
His goal was to show that sugar (i.e. snacks and soda) is not in any way worse than general carbs (i.e. bread and rice), so there is no need to tax or ban Mars and Coca Cola (which happen to be his sponsors).
Some of the flaws of the studies he uses: 1) They used pure fructose (which is very poorly absorbed); 2) They counted HFCS as “carb” and concluded sugar is not worse; 3) They used mice studies, even though they metabolize fructose / uric acid very differently than humans do. 4) They neglected that fructose is appetite-stimulating / suppresses satiety, thus contributes to over-consumption in ad lib; 5) They didn’t distinguish between soda and fruit; and many other fundamental flaws.
Overall, the Sievenpieper studies are not reliable in any way.
Hi Andrew,
I fail to see how Cuba doesn’t support my argument. I’m saying that calorie intake and physical activity trump the fattening effects of sugar. That is precisely what the Cuba evidence shows.
Regarding the Sievenpiper meta-analyses, you seem to believe that sugar is fattening beyond its calorie content. Can you cite evidence to support that position? All evidence I’m aware of, including the metabolic ward studies, suggest that is incorrect.
Hi Stephan,
1) if your argument is that a 1000cal reduction leads to weight loss no matter what you eat, that’s trivial. But that was not your point in the podcast: you said Cuban sugar consumption went up (which we don’t even know!) and yet they lost weight. It is actually quite possible that their absolute refined sugar consumption went down (total non-fruit or as beverages).
2) There are many, many studies that show fructose/sugar has unique effects on weight, liver, fat metabolism, insulin, leptin, cholesterol composition, cancer growth, and many other key indicators. If you’re really not aware of these, that’s frankly rather shocking. Also, if you haven’t been aware of the serious flaws of the Sievenpieper meta-analyses, that’s also not a good sign.
Just a few examples:
http://care.diabetesjournals.org/content/early/2012/08/24/dc12-0540
https://academic.oup.com/ajcn/article/86/4/1174/4649619
https://idmprogram.com/fructose-causes-insulin-resistance-hormonal-obesity-xxxii/
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2682989/
https://www.ethz.ch/en/news-and-events/eth-news/news/2015/06/fructose-vicious-circle.html
https://academic.oup.com/ajcn/article/76/5/911/4689540
https://www.reuters.com/article/us-fructose-diabetes/clues-seen-to-how-fructose-may-promote-diabetes-idUSTRE52254920090303
See also: https://onlinelibrary.wiley.com/doi/abs/10.1002/oby.21371
Moreover, weight is just one indicator to overall metablic health.
Here’s a Cuban ration list from 1994:
“Sylvia and Hilda get one bread roll each per day. Milk or chicken are not available to adults. Hilda is allowed a litre of milk every two days and a quarter piece of chicken a month. (In fact, chicken has not appeared for more than two months).
They are allowed half a dozen eggs each a month. The only meat is part of a soya and mincemeat mixture but each is allowed only half a pound a month. Their other monthly rations are: rice – five pounds per person; black beans (a Cuban staple) – one pound per person; sugar – five pounds; salt – one ounce; rum – half a bottle. ”
The sugar ration works out to 75 grams a day, or 300 calories, which would be 20% of a 1500 calorie per day diet, but it appears that they were eating well below that because the rice ration is the same size as the sugar ration.
https://www.independent.co.uk/news/world/millions-of-cubans-facing-starvation-hunger-is-fuelling-an-exodus-of-desperate-refugees-writes-phil-1417691.html
.17 Declining Sugar Consumption
It’s obvious you cherry picked articles from ncbi. There are articles concluding sugar intake rose:
https://www.ncbi.nlm.nih.gov/pubmed/19056548
https://www.ncbi.nlm.nih.gov/pubmed/18519465
https://www.ncbi.nlm.nih.gov/pubmed/26244332
Sugary drinks and desserts are easily available at every corner more than ever before and business is booming.
It’s pure delusion to believe people are consuming less sugar in developed countries.
Hi Bob,
If you read the abstracts of the studies you cited, you’ll see that they are consistent with what I wrote above. What the graph shows is that sugar intake rose until 1999, then declined thereafter. That’s consistent with the studies you cited.
Stephan,
Not quite. The data you reference do not actually monitor how much sugar people put in their bodies; it measures “estimated deliveries for domestic food and beverage use” and divides it by the population.
Your point 17 is misleading because it doesn’t use actual intake, it uses deliveries and then arbitrarily uses a estimate of consumption.
Hi Bob,
Again, I recommend reading more carefully. If you look at all the references I cite under point 17, you will see that they represent several different methods for estimating sugar intake that all report the same decline. Some of the methods are based on production, some are based on self-reported intake, but they all say the same thing. I’m sorry, but you are simply incorrect.
Just a note here that I’m not publishing any more of Bob Plant’s comments due to a refusal to accurately represent the references he cites. You can see evidence of that by examining the references he cited above. People can disagree with me freely here, as long as it’s respectful, but I’m not going to publish comments that are obviously misleading.
Hi Stephen,
Have your seen/read the website/book from Cian Foley, Don’t Eat for Winter? https://www.donteatforwinter.com/
I recently found his blog and find the information there fascinating, and it rings very true to me. What he promotes sounds like what you’re saying. I am a perimenopausal woman that has been struggling to keep my weight down at this point in my life, and his approach to eating/weight loss is finally making a dent in my weight loss efforts.
What do you think of this approach? Has anybody else tried it? Has it worked for you, especially long term? I’m especially interested to hear from other peri/menopausal women that may have tried this, or something similar, and had results where nothing else was working. I’m happy to hear from anyone though!
Hi Stephen,
Have you had a chance to look at the link above? I’m wondering what you think about it. Thanks.
I watched the whole show.
A quick point re your delivery – I thought you started off a bit high handed but it quickly became evident that Gary was only interested in a monologue, not a discussion. His repeated attempts to interrupt should have been policed more by Rogan and I felt he got more “air time”. Gary just can’t make a succinct point.
There was no talk of epigenetics and how starvation may alter the bodies attempts to store fat and if this may mean both approaches have some validity (I am not a scientist and know nothing of the area). Could it also explain why the inheritance issues you were discussing.
You need to go on alone and never agree to share the platform with Gary without a very strong moderator.
Hi Stephan,
I really enjoyed the exchanges you had with Gary. It’s a good thing that you did not let him talk over you. Trying to enforce good manners is not arrogant. Thanks for this update. Cheers!
I didn’t listen to the podcast, but this is a great annotated bibliography all on its own. I really appreciate your work in gathering all of this information. I’ve read your blog here for years.
I started watching the debate thinking you’d be arguing the energy balance model, and you did, but you also brought in this argument about the brain being the regulator. I hadn’t heard it put that way before. Very interesting. I’ve just ordered your book.
I run a site for naturally skinny guys who are trying to gain weight. I was wondering if you knew where I could look if I was trying to figure out how to eat MORE calories (to gain muscle, mind you). Could I use the ideas in your book in reverse in order to become MORE hungry?
Thanks for the entertaining debate. It was bordering on the unfair, so badly Taubes was prepared for this. There are couple of things that could be added to the debate. In several studies ingestion of protein elicited stronger insulin response than ingestion of dextrose (yet protein rich diet is a good guy). Also, insulin is anorexogenic, which means that palatability of carbs is n to due to insulin.
Anyway, the debate was the closes it could get to a good UFC match. Thanks again!